Is oxidation and rust the same thing

David Jones
Published Apr 15, 2026
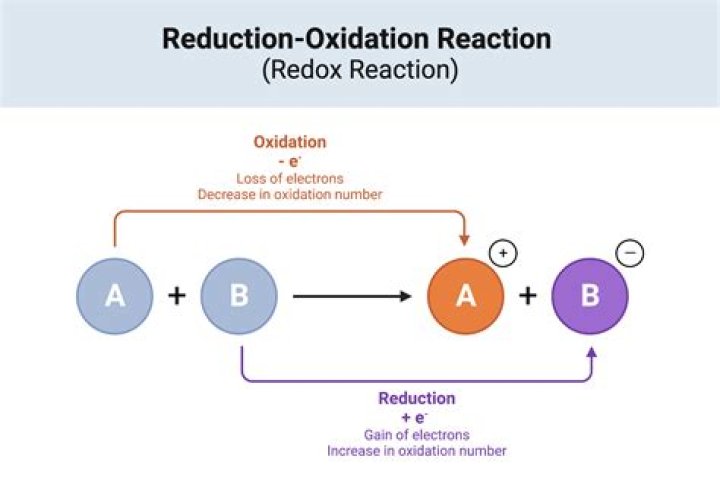
Firstly, is oxidation and rust the same thing? Oxidation is the general chemical reaction of losing one or more electrons in an atom, molecule or ion, however, rust is an oxidation reaction only when iron reacts with water and oxygen.
Is rust also known as oxidation?
Rusting is an oxidation reaction. The iron reacts with water and oxygen to form hydrated iron(III) oxide, which we see as rust.
What is rust also known as?
Rust is also known as iron oxide and corrosion. This chemical compound comes in many different combinations of iron and oxygen.
What is the same as oxidation?
Corrosion is deterioration or destruction of metals as a result of rain, sleet, snow, etc. and oxidation is an electrochemical breakdown of the metal. Oxygen leads to oxidation. … Corrosion mainly happens to metals whereas oxidation can take place anywhere.Do other metal corrode or oxidize?
All metals, with the exception of the precious metals, will oxidize when exposed to oxygen and an electrolyte (i.e. atmospheric moisture).
What is meant oxidation?
Oxidation is a process in which a chemical substance changes because of the addition of oxygen. The reaction between magnesium metal and oxygen involves the oxidation of magnesium. … Oxidation is a process in which a chemical substance changes because of the addition of oxygen.
Why is rust called rust?
TL;DR: Rust is named after a fungus that is robust, distributed, and parallel. It is also a substring of “robust”.
What is copper rust?
Copper corrosion is the corrosion of materials made of copper or copper alloys. When exposed to the atmosphere, copper oxidizes, causing normally bright copper surfaces to tarnish. After a few years, this tarnish gradually changes to dark brown or black, and finally to green.Is corrosion always oxidation?
Corrosion and oxidation are two different terms express the same idea. Oxidation reaction is one of the two simultaneous reactions of redox reactions. Corrosion is a type of oxidation. Corrosion happens when the metal atoms on a metal surface get oxidized in the presence of oxygen and water.
Is rust and ferric oxide same?Ferric oxide is sometimes called rust because it shares physical and chemical composition properties similar to rust or corrosion. The chemical structure of ferric oxide has iron (Fe) in the center bound to six oxygen ligands.
Article first time published onIs rust hydrated ferric oxide?
Rust is a reddish-brown oxide formed by the reaction of iron and oxygen in the presence of water or air moisture. … Rust consists of hydrated iron(III) oxides Fe2O3 .
Is rust a ferrous oxide?
Technically rust is Hydrated Iron (III) Oxide, also known as iron oxide (Fe²O³), as it is caused when iron reacts with oxygen and water – this reaction is known as oxidizing.
What is the difference between rusting and corrosion?
Corrosion is the process by which certain materials, metals and non-metals, deteriorate as a result of oxidation. Rusting is oxidation of iron in the presence of air and moisture. Corrosion can occur on materials such as ceramics or polymers. Rusting occurs on surfaces of iron and its alloys.
What metal does not rust?
Platinum, gold & silver Known as the precious metals, platinum, gold and silver are all pure metals, therefore they contain no iron and cannot rust. Platinum and gold are highly non-reactive, and although silver can tarnish, it is fairly corrosion-resistant and relatively affordable by comparison.
Does aluminum rust or corrode?
Rust is a type of corrosion (the wearing-away of metal), and to put it simply, aluminium does not rust, but it does corrode. Although these terms are often used interchangeably, they are fundamentally different. As with any metal, when it comes into contact with oxygen, an oxide layer will form on aluminium.
Is Rust built on C?
It never was primarily written in C or C++, though LLVM is used in today’s compiler. Originally it was OCaml, and then it was ported to Rust. From the posts I see Rust was written in OCaml and then Rust.
Can metal Rust underwater?
Corrosion is also accelerated in pipes carrying sea water travelling at high velocities. Water is H2O hydrogen and oxygen, Rust is iron oxide, the result you get from exposing iron to oxygen, called oxidation. In conclusion the iron will rust underwater, as water is composed of oxygen.
Is Rust as fast as C?
As far as speed/performance, Rust is on the same page as C++. There are situations where it is easier to write faster programs in C++ because it’s easy to ignore fundamental problems in the programs.
What are the examples of oxidation?
The iron metal is oxidized to form the iron oxide known as rust. Electrochemical reactions are great examples of oxidation reactions. When a copper wire is placed into a solution that contains silver ions, electrons are transferred from the copper metal to the silver ions. The copper metal is oxidized.
Why is it called oxidation?
The term oxidation was first used by Antoine Lavoisier to signify the reaction of a substance with oxygen. Much later, it was realized that the substance, upon being oxidized, loses electrons, and the meaning was extended to include other reactions in which electrons are lost, regardless of whether oxygen was involved.
What weathering is oxidation?
Oxidation is another kind of chemical weathering that occurs when oxygen combines with another substance and creates compounds called oxides. … When rocks, particularly those with iron in them, are exposed to air and water, the iron undergoes oxidation, which can weaken the rocks and make them crumble.
What is the chemical name for rust?
Rust is the common name for a very common compound, iron oxide. Iron oxide, the chemical Fe2O3, is common because iron combines very readily with oxygen — so readily, in fact, that pure iron is only rarely found in nature.
Is rust a precipitate?
Iron(II) hydroxide is precipitated. Rust is then quickly produced by the oxidation of the precipitate. The rusting of unprotected iron in the presence of air and water is then inevitable because it is driven by an electrochemical process.
Does brass oxidize?
Copper, Bronze and Brass Copper oxidizes over time to form a green patina, which actually protects the metal from further corrosion. … Brass is an alloy of copper, zinc, and other elements, which also resists corrosion.
Is zinc a rust?
The short to the question is, yes. Zinc does rust. Like all metals, zinc corrodes when exposed to air and moisture. However, this element does not rust like most other metals.
Is steel a rust?
Rust is an iron oxide, a usually reddish-brown oxide formed by the reaction of iron and oxygen in the catalytic presence of water or air moisture. … Rusting is the common term for corrosion of elemental iron and its alloys such as steel.
Does copper get rusted?
Copper will never rust for the same reason as bronze — it contains too little iron. Though it will not rust, copper can form a green film, or patina, on its surface over time. … Her copper skin originally looked brown, but it has turned green over time due to the copper’s oxidation.
Is rust on metal a fungus?
Rusts are plant diseases caused by pathogenic fungi of the order Pucciniales (previously known as Uredinales). An estimated 168 rust genera and approximately 7,000 species, more than half of which belong to the genus Puccinia, are currently accepted.
How does iron rust?
Rust is the result of corroding steel after the iron (Fe) particles have been exposed to oxygen and moisture (e.g., humidity, vapor, immersion). … Oxygen causes these electrons to rise up and form hydroxyl ions (OH). The hydroxyl ions react with the FE⁺⁺ to form hydrous iron oxide (FeOH), better known as rust.
What is Fe2O3 called?
Ferric Oxide (fe2o3)
What makes rust different from iron?
Its scientific name is iron oxide (Fe2O3). Rust forms when iron and oxygen react in the presence of water or moisture in the air. … Rust occurs when iron or its alloys, such as steel, corrode. The surface of a piece of iron will corrode first in the presence of oxygen and water.