Is Naphthalene a pure substance

John Castro
Published Apr 15, 2026
determination of a melting point, eg for naphthalene (pure substance) or candle wax (impure substance) separation of liquids by distillation, eg ethanol from water, and by paper chromatography.
Is naphthalene A mixture or pure substance?
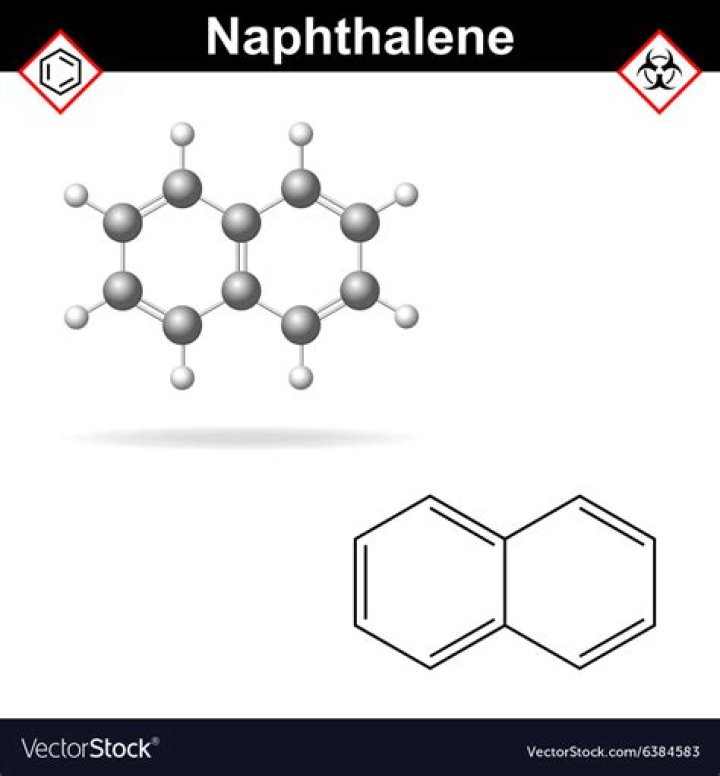
Naphthalene is a specific organic compound: a polynuclear aromatic hydrocarbon of formula C10H8 with a chemical structure that can be envisioned as two benzene rings fused together.
Is naphthalene an element compound or mixture?
Explanation: When two or more different elements chemically combine together then it results in the formation of a compound. Chemical formula of naphthalene is . Naphthalene is composed of carbon and hydrogen atoms, therefore, it is a compound.
Is naphthalene a mixture?
Napthalene ball is compound. … It is the simplest of fused or condensed rings hydrocarbon compounds composed of two benzene rings sharing two adjacent carbon atoms as shown in figure.What type of compound is naphthalene?
Naphthalene is an organic compound with formula C10H8. It is the simplest polycyclic aromatic hydrocarbon, and is a white crystalline solid with a characteristic odor that is detectable at concentrations as low as 0.08 ppm by mass.
Is naphthalene a structure?
It is the simplest polycyclic aromatic hydrocarbon, and is a white crystalline solid with a characteristic odor that is detectable at concentrations as low as 0.08 ppm by mass. As an aromatic hydrocarbon, naphthalene’s structure consists of a fused pair of benzene rings.
Is naphthalene a liquid?
Naphthalene (NAF-thuh-leen) is a white crystalline volatile solid with a characteristic odor often associated with mothballs. The compound sublimes (turns from a solid to a gas) slowly at room temperature, producing a vapor that is highly combustible.
Is naphthalene soluble or insoluble?
Naphthalene is a nonpolar compound. Thus, it is insoluble in highly polar solvents like water.Is naphthalene molecular or ionic?
Naphthalene is a molecular solid. If the crystals contains only individuals atoms; as in solid argon or krypton or if they are composed of non polar molecules as in naphthalene, the only attraction between the molecules are the London forces. Name an important source from which naphthalene balls are obtained.
Is naphthalene a neutral compound?Naphthalene, on the other hand, is a neutral organic compound, and therefore is not soluble in water or the aqueous solutions that will be produced throughout the experiment.
Article first time published onIs naphthalene a heterocyclic compound?
The first example is azulene, a blue-colored 10 π-electron aromatic hydrocarbon isomeric with naphthalene. The second and third compounds are heterocycles having aromatic properties. Pyridine has a benzene-like six-membered ring incorporating one nitrogen atom.
Why is naphthalene an aromatic compound?
As we are getting a whole number from, 4nπ+2 formula for naphthalene, so naphthalene is aromatic. Therefore, the naphthalene is an aromatic compound according to Huckel’s rule because it has 4nπ+2 delocalised electrons. Note: Pi bonds are known as delocalized bonds. Pi bonds cause the resonance.
Is naphthalene saturated or unsaturated?
Camphor and naphthalene unsaturated and alcohol is saturated. Naphthalene =unsaturated.
Is naphthalene a base?
The pKa’s of benzoic acid and 2-naphthol are 4.17 and 9.5, respectively, while naphthalene is a neutral compound. Since benzoic acid is much more acidic than 2-naphthol, the weak base, sodium bicarbonate, will be able to effectively remove benzoic acid’s acidic hydrogen.
Is naphthalene aromatic or not?
Naphthalene, with two fused rings, is the simplest polycyclic aromatic molecule. Note that all the carbon atoms except those at the points of fusion have a bond to a hydrogen atom. Naphthalene, which has 10 π electrons, satisfies the Hückel rule for aromaticity.
Is naphthalene a non Benzenoid aromatic compound?
Examples of benzenoid aromatic compounds include naphthalene, anthracene, phenanthrene, aniline etc. A non benzenoid aromatic compound contains conjugated pi electron systems with a ring of 5 to 7 carbon atoms.
Is naphthalene an atom?
naphthalene, the simplest of the fused or condensed ring hydrocarbon compounds composed of two benzene rings sharing two adjacent carbon atoms; chemical formula, C10H8.
What type of solid is naphthalene?
Naphthalene is a crystalline solid.
Is naphthalene a hazardous substance?
Naphthalene is a COMBUSTIBLE SOLID. It may also be transported in a “molten” or heated form. The vapor given off when Naphthalene is heated is FLAMMABLE and a DANGEROUS FIRE HAZARD.
What are the properties of naphthalene?
- Naphthalene exists in crystalline form.
- Naphthalene is generally white in colour. …
- The molecular weight of naphthalene is 128.18 g/mol.
- Naphthalene is insoluble in water at a normal room temperature.
- Naphthalene has an aromatic odour.
- The vapour pressure of naphthalene is 0.087 mmHg.
Is naphthalene an electrolyte?
Molten lead(II) bromide, sodium hydroxide solution and copper(II) sulphate solution are electrolytes whereas molten acetamide, molten naphthalene and glucose solution are non-electrolytes.
Is NH4NO3 ionic or molecular?
Ionic bond. NH4NO3 is a nitrate salt of the ammonium cation. Since ammonium is a cation and bonds with the anion nitrate, hence the compound is bonded by an ionic bond.
Can naphthalene sublime?
Naphthalene is a solid that sublimes at standard atmospheric temperature with the sublimation point at around 80 °C or 176 °F. At low temperature, its vapour pressure is high enough, 1 mmHg at 53 °C, to make the solid form of naphthalene evaporate into gas.
Why naphthalene is non-polar?
Naphthalene is a non-polar molecule. This is because a bond between carbon and hydrogen has an electronegativity value of 0.4 and naphthalene consists…
Is naphthalene a polar solvent?
Naphthalene is a polar nonpolar substance. As a result, it’s insoluble in polar solvents like water. Since non-polar solutes cannot deal with the intense affinity that polar solvent molecules have with each other, they do not dissolve in polar solvents like water.
Is naphthalene is soluble in benzene?
It melts at 80°C, boils at 218°C, and sublimes upon heating. It is insoluble in water, somewhat soluble in ethanol, soluble in benzene, and very soluble in ether, chloroform, or carbon disulfide. Naphthalene is obtained from coal tar, a byproduct of the coking of coal.
Is naphthalene a polar or nonpolar compound?
naphthalene is a non polar substance so it will dissolve in non poalr solvent and camphor pon the other hand will dissolve in polar solvent.
Is naphthalene same as camphor?
As nouns the difference between camphor and naphthalene is that camphor is (organic compound) a white transparent waxy crystalline isoprenoid ketone, with a strong pungent odour, used in pharmacy while naphthalene is a white crystalline hydrocarbon manufactured from coal tar; used in mothballs.
Is naphthalene aromatic or aliphatic?
Naphthalene is the simplest polycyclic aromatic hydrocarbon since it is only a bicyclic molecule made up of two aromatic benzenes.
What type of bonding is present in naphthalene?
The naphthalene ion forms unconventional carbon-based ionic hydrogen bonds with H2O and CH3OH (CH delta+center dot center dot center dot OH2 and CH delta+center dot center dot center dot OHCH3, respectively) which can be extended to hydrogen bonding chains with additional solvent molecules.
Is benzene heterocyclic compound?
In their general structure, heterocyclic compounds resemble cyclic organic compounds that incorporate only carbon atoms in the rings—for example, cyclopropane (with a three-carbon-atom ring) or benzene (with a six-carbon-atom ring)—but the presence of the heteroatoms gives heterocyclic compounds physical and chemical …