What is the unit of atomic mass called

John Castro
Published Mar 29, 2026
Atomic weight is measured in atomic mass units (amu), also called daltons. See below for a list of chemical elements and their atomic weights.
What is the unit of atomic mass called and what is its symbol?
The dalton or unified atomic mass unit (symbols: Da or u) is a unit of mass widely used in physics and chemistry. It is defined as 1⁄12 of the mass of an unbound neutral atom of carbon-12 in its nuclear and electronic ground state and at rest.
Are Daltons and amu the same?
The dalton is simply a renaming of the unified atomic mass unit, so they have equal values—there is no distinction between the two units other than name and symbol, so the two names are synonymous as are the two unit symbols.
Is atomic mass unit an SI unit of atomic mass?
The unified atomic mass unit (unit symbol: u) is a non-SI unit of mass, defined as one-twelfth the mass of a single 12C atom in its ground state. … The unit symbol “mmu,” meaning a millimass unit, is also an appropriate unit in SI. Instead of “mmu,” “mDa” or and “10-3 u” should be used.Is amu and u the same?
In chemistry, an atomic mass unit or AMU is a physical constant equal to one-twelfth of the mass of an unbound atom of carbon-12. It is a unit of mass used to express atomic masses and molecular masses. … The symbol for the unit is u (unified atomic mass unit) or Da (Dalton), although AMU may still be used.
What does amu stand for?
abbreviation. atomic mass unit (def.
Is amu same as g mol?
The mass of one mole of atoms of a pure element in grams is equivalent to the atomic mass of that element in atomic mass units (amu) or in grams per mole (g/mol). Although mass can be expressed as both amu and g/mol, g/mol is the most useful system of units for laboratory chemistry.
Is Slug a unit of mass?
The slug is a derived unit of mass in a weight-based system of measures, most notably within the British Imperial measurement system and the United States customary measures system. … One slug is a mass equal to 32.1740 lb (14.59390 kg) based on standard gravity, the international foot, and the avoirdupois pound.How do you use amu?
The AMU is used to express the relative masses of, and thereby differentiate between, various isotopes of elements. Thus, for example, uranium-235 (U-235) has an AMU of approximately 235, while uranium-238 (U-238) is slightly more massive.
Which symbol replaces the unit of atomic mass amu?Presently the new symbol used is ‘u’ (known as unified mass) in place of amu.
Article first time published onWhat is the value of 1 amu in Gram?
1 amu is equal to 1.66 × 10^-24 g.
Is a neutron 1 amu?
neutron: A subatomic particle forming part of the nucleus of an atom. It has no charge. It is equal in mass to a proton or it weighs 1 amu.
What is the Amu of a neutron?
mn = mass of a neutron (1.008665 amu)
How many amu is a proton?
The masses of protons and neutrons are nearly equal (1.0073 and 1.0087 amu respectively).
What is atomic mass unit class 11?
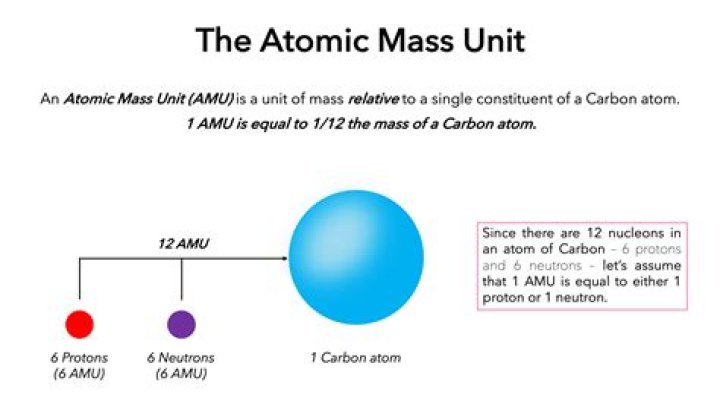
One atomic mass unit is defined as a mass exactly equal to one – twelfth the mass of one carbon -12 atom. It is used to express the mass of atomic and subatomic particles. Because the atomic mass of an atom is actually very-very small because atoms are extremely small.
How do you convert AMU to joules?
The formula used in unified atomic mass units to joules conversion is 1 Unified Atomic Mass Unit = 1.4924179527347E-10 Joule.
What is the atomic mass of carbon 🔹?
On the periodic table the mass of carbon is reported as 12.01 amu. This is the average atomic mass of carbon.
Why is carbon-12 the standard?
Since carbon forms millions of compounds, carbon is a good starting point. Molar mass divided by Avagadro’s number is atomic mass, especially if you are dealing with single isotopes. Again, carbon 12 is stable and easily available, so is used as a standard.
Is Barn a unit of mass?
BarnUnit systemparticle physicsUnit ofareaSymbolbNamed afterthe broad side of a barn
Is lbf the same as LB?
“Lbf” refers to the gravitational force placed by a matter on the Earth’s surface, while “lb” deals with the measurement of force. A pound force equals the product of 1 pound and the gravitational field. “Lb” and “lbf” are basically similar with each other since they both involve the same force.
Is joules a unit of force?
joule, unit of work or energy in the International System of Units (SI); it is equal to the work done by a force of one newton acting through one metre. Named in honour of the English physicist James Prescott Joule, it equals 107 ergs, or approximately 0.7377 foot-pounds.
What 1u means?
1-An atomic mass unit (u) is a unit of mass used to express atomic and molecular weights. One atomic mass unit (1u) or 1 a.m.u. is defined as one twelfeth (1/12) of the mass of an atom of carbon-12.
What is the mass of hydrogen in terms of AMU?
The mass of a hydrogen molecule is 2.016 amu.
Is an electron 1 amu?
Protons, neutrons, and electrons: Both protons and neutrons have a mass of 1 amu and are found in the nucleus. … Electrons have a mass of approximately 0 amu, orbit the nucleus, and have a charge of -1.
How is atomic mass unit calculated?
For any given isotope, the sum of the numbers of protons and neutrons in the nucleus is called the mass number. This is because each proton and each neutron weigh one atomic mass unit (amu). By adding together the number of protons and neutrons and multiplying by 1 amu, you can calculate the mass of the atom.
What has a mass of 1 amu?
Since the nucleus accounts for nearly all of the mass of the atom, a single proton or single neutron has a mass of approximately 1 amu.
What is the unit of proton?
proton, stable subatomic particle that has a positive charge equal in magnitude to a unit of electron charge and a rest mass of 1.67262 × 10−27 kg, which is 1,836 times the mass of an electron.
What is the mass of proton and electron?
ParticleSymbolMass (amu)protonp+1electrone−5.45 × 10−4neutronn01
What is electron and proton and neutron?
Atoms are made of extremely tiny particles called protons, neutrons, and electrons. Protons and neutrons are in the center of the atom, making up the nucleus. Electrons surround the nucleus. Protons have a positive charge. Electrons have a negative charge.
What is the atomic mass of an electron?
The electron has a mass of about 9.109382911 × 10−28 gram. The mass of a proton or neutron is about 1,836 times larger. This explains why the mass of an atom is primarily determined by the mass of the protons and neutrons in the nucleus. The electron has other intrinsic properties.
What is proton and neutron?
Protons are a type of subatomic particle with a positive charge. Protons are bound together in an atom’s nucleus as a result of the strong nuclear force. Neutrons are a type of subatomic particle with no charge (they are neutral).



