What is the molecular geometry of a compound

David Jones
Published Apr 19, 2026
Molecular geometry, also known as the molecular structure, is the three-dimensional structure or arrangement of atoms in a molecule. Understanding the molecular structure of a compound can help determine the polarity, reactivity, phase of matter, color, magnetism, as well as the biological activity.
How do you find the geometry of a compound?
- VSEPR Rules:
- Identify the central atom.
- Count its valence electrons.
- Add one electron for each bonding atom.
- Add or subtract electrons for charge (see Top Tip)
- Divide the total of these by 2 to find the total.
- number of electron pairs.
- Use this number to predict the shape.
What is geometry of simple compounds?
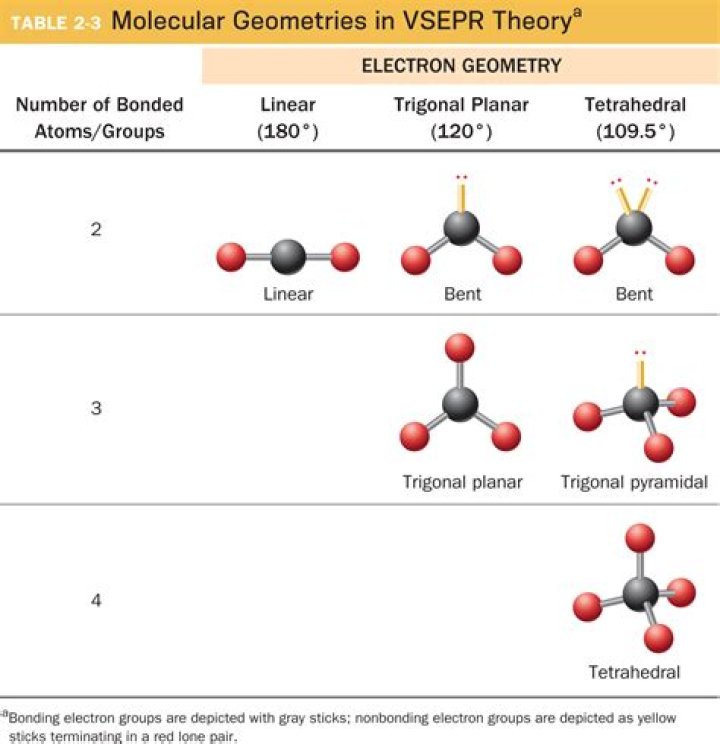
Molecular Geometries. The VSEPR theory describes five main shapes of simple molecules: linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral.
What is molecular geometry example?
For example; four electron pairs are distributed in a tetrahedral shape. … If there is one lone pair of electrons and three bond pairs the resulting molecular geometry is trigonal pyramidal (e.g. NH3). If there are two bond pairs and two lone pairs of electrons the molecular geometry is angular or bent (e.g. H2O).What is the molecular geometry of a molecule with 2 outer atoms and 1 lone pair on the central atom?
Nathan L. This is a bent molecule.
What is the molecular geometry of h2o?
Water has 4 regions of electron density around the central oxygen atom (2 bonds and 2 lone pairs). These are arranged in a tetrahedral shape. The resulting molecular shape is bent with an H-O-H angle of 104.5°.
What is molecular geometry vs electron geometry?
The molecular geometry definition in chemistry is the arrangement of atoms in relation to a central atom in three-dimensional space. Electron geometry is the arrangement of electron groups.
What are the 6 basic molecular shapes?
The 6 basic molecular shapes are linear, trigonal planar, angular (bent), tetrahedral, trigonal pyramidal, and trigonal bipyramidal.What is the molecular geometry of nh3?
The ammonia molecule has a trigonal pyramidal shape with the three hydrogen atoms and an unshared pair of electrons attached to the nitrogen atom.
What is the molecular geometry of a molecule with 4 outer atoms and 1 lone pairs on the central atom?# of bonding groups/domains on ‘central’ atom# of lone pair electrons on ‘central’ atomMolecular Geometry40tetrahedral31trigonal pyramidal22bent50trigonal bipyramidal
Article first time published onWhat is the molecular geometry of a molecule with 5 outer atoms and 1 lone pairs on the central atom?
Thus, the molecular geometry of the molecule with 5 outer atoms and 1 lone pair in the central atom is square pyramidal.
What is the molecular geometry of no3?
NO3– Molecular Geometry And Bond Angles In essence, nitrate has 3 electron domains and no lone pairs. Therefore, NO3– molecular geometry is slightly bent and is trigonal planar. The bond angle is 120o.
What's the molecular geometry of SO2?
Name of moleculeSulfur dioxideMolecular Geometry of SO2Trigonal planarHybridization of SO2sp2 hybridizationNo of Valence Electrons in the molecule18
What is the molecular geometry of CH3CN?
Name of MoleculeAcetonitrile/methyl cyanideChemical formulaCH3CNMolecular geometry of CH3CNTetrahedral/linearElectron geometry of CH3CNTetrahedral/linearHybridizationSp³ and Sp
What is the molecular geometry of HCN?
Hydrogen cyanide has linear molecular geometry with bond angles of 180 degrees. As hydrogen and nitrogen tend to be far from each other, HCN forms a linear shape.
What is octahedral molecular geometry in chemistry?
In chemistry, octahedral molecular geometry describes the shape of compounds with six atoms or groups of atoms or ligands symmetrically arranged around a central atom, defining the vertices of an octahedron. The octahedron has eight faces, hence the prefix octa.
What is the molecular geometry of a molecule with 3 outer atoms and 2 lone pair on the central atom?
The shape of a molecule is based on its molecular geometry: when determining molecular geometry, atoms and lone pairs are treated differently. When there are 3 atoms and 2 lone pairs around the central atom, it corresponds to AX3E2 or T-shaped. The molecular geometry of the molecule being described is T-shaped.
What is the molecular geometry of a molecule with 4 groups attached to the central atom?
A molecule with four electron groups about the central atom orients the four groups in the direction of a tetrahedron, as shown in Figure 9.3 “Tetrahedral Geometry”. If there are four atoms attached to these electron groups, then the molecular shape is also tetrahedral.
What is the molecular structure of SnCl − 3?
SOLUTION (a) The Lewis structure for the SnCl3-; ion is: The central Sn atom is surrounded by one nonbonding electron pair and three single bonds. Thus, the electron-pair geometry is tetrahedral with three of the corners occupied by the bonding pairs of electrons.
Is bent molecular geometry?
In chemistry, molecules with a non-collinear arrangement of two adjacent bonds have bent molecular geometry, also known as angular or V-shaped. … Water (H2O) is an example of a bent molecule, as well as its analogues. The bond angle between the two hydrogen atoms is approximately 104.45°.
What is the electron geometry of brf5brf5?
BrF5 Molecular Geometry And Bond Angles BrF5 molecular geometry is said to be square pyramidal with a bond angle of 90o each.
What is the molecular geometry of cli5 CLI 5?
Since there are six electron pairs on the central atom, then VSEPR theory tells us that the electron pair geometry is octahedral and with 5 bonding pairs and 1 lone pair, the molecular geometry is square pyramidal.
What is electron-pair geometry?
electron-pair geometry: arrangement around a central atom of all regions of electron density (bonds, lone pairs, or unpaired electrons) linear: shape in which two outside groups are placed on opposite sides of a central atom. molecular structure: structure that includes only the placement of the atoms in the molecule.
What is electronic geometry in chemistry?
The electronic geometry is an idea of where the regions of electron density are surrounding the central atom. A region of electron density can either be bonding or non-bonding.



