What is the function of trypsin

Mia Russell
Published Mar 30, 2026
Trypsin is an enzyme that helps us digest protein. In the small intestine, trypsin breaks down proteins, continuing the process of digestion that began in the stomach. It may also be referred to as a proteolytic enzyme, or proteinase. Trypsin is produced by the pancreas in an inactive form called trypsinogen.
What is the function of pepsin and trypsin?
Function: Pepsin acts on proteins and converts them into peptones, while trypsin converts peptones into polypeptides.
What is the function of lipase and trypsin?
Amylase digests carbohydrates, lipase digests fats, and trypsin digests proteins. The pancreas also secretes large amounts of sodium bicarbonate, which protects the duodenum by neutralizing the acid that comes from the stomach.
What are the functions of trypsin Class 10?
Trypsin, a serine protease is an enzyme that helps us in digesting protein. It continues the process of digestion that began in the stomach in the small intestine by breaking down proteins. This enzyme is produced by the pancreas in an inactive form called trypsinogen.What organ is trypsin?
It has long been known that trypsin is produced as a zymogen (trypsinogen) in the acinar cells of the pancreas, is secreted into the duodenum, is activated into the mature form of trypsin by enterokinase, and functions as an essential food-digestive enzyme.
What is the means of trypsin?
Definition of trypsin : a proteolytic enzyme that is secreted in the pancreatic juice in the form of trypsinogen, is activated in the duodenum, and is most active in a slightly alkaline medium.
What is the function of trypsin and pepsin Class 10?
The trypsinogen enters the small intestine through the common bile duct and is converted to active trypsin. This active trypsin acts with the other two principal digestive proteinases — pepsin and chymotrypsin — to break down dietary protein into peptides and amino acids.
What are the two functions of hydrochloric acid?
Chief function of HCl is (1) To maintain low pH to promote growth of microorganisms (2) To facilitate absorption (3) To maintain low pH to activate pepsinogen to form pepsin (4) To dissolve enzymes secreted in stomach. Hydrochloric acid is an essential acid in the stomach.What is the function of aminopeptidase?
Aminopeptidases catalyze the cleavage of amino acids from the amino terminus of protein or peptide substrates. They are widely distributed throughout the animal and plant kingdoms and are found in many subcellular organelles, in cytoplasm, and as membrane components.
What is the difference between trypsin and chymotrypsin?Selection. The main difference between chymotrypsin and trypsin is the amino acids they select for. Chymotrypsin is the enzyme that selects for the aromatic amino acids: phenylalanine, tryptophan, and tyrosine. Trypsin is the enzyme that selects for the basic amino acids: lysine and arginine.
Article first time published onWhat is the function of trypsin and lipase class 10?
Amylase digests carbohydrates, lipase digests fats, and trypsin digests proteins. The pancreas also secretes large amounts of sodium bicarbonate, which protects the duodenum by neutralizing the acid that comes from the stomach.
What is the main function of lipase class 10?
Lipase enzyme breaks down dietary fats into smaller molecules known as glycerol and fatty acids. A little quantity of lipase, known as gastric lipase is produced by the cells of the stomach. This enzyme mainly digests fat present in the food.
What is the function of salivary amylase and trypsin?
Salivary amylase- It is secreted by the salivary gland and it acts on starch present in the food and breaks it down into smaller carbohydrate molecules. d. Trypsin-Trypsin secreted by the pancreas, hydrolyses more protein present in food that isn’t broken down by pepsin earlier.
What's the function of trypsin?
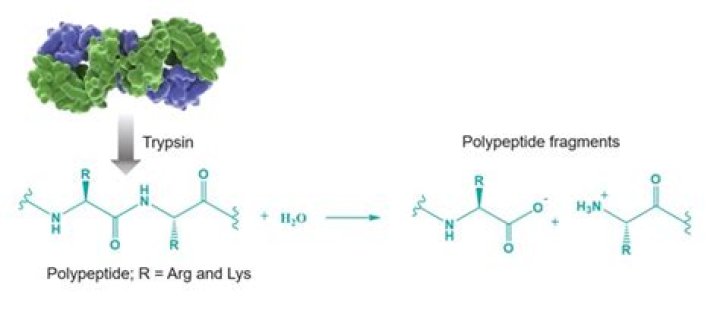
Function. In the duodenum, trypsin catalyzes the hydrolysis of peptide bonds, breaking down proteins into smaller peptides. The peptide products are then further hydrolyzed into amino acids via other proteases, rendering them available for absorption into the blood stream.
What is the role of trypsin in cell culture?
When added to a cell culture, trypsin breaks down the proteins which enable the cells to adhere to the vessel. … Trypsin, an enzyme commonly found in the digestive tract, can be used to “digest” the proteins that facilitate adhesion to the container and between cells.
What is the function of trypsin and chymotrypsin?
Trypsin (also sometimes referred to as a proteinase) goes to work with two other proteinases called pepsin and chymotrypsin to break down protein (from food) into amino acids. Amino acids are building blocks of protein and they are used in the body for many functions, including: Producing hormones.
What is the difference between trypsin and lipase?
Trypsin and lipase are both digestive enzymes with rather dissimilar properties and structure. Trypsin is a protease and catalyzes hydrolysis of proteins while lipase catalyzes hydrolysis of lipids.
What is difference between trypsin and pepsin?
It is an enzyme which is secreted by the stomach. It is an enzyme secreted by pancreas. … The one point difference between pepsin and trypsin is that pepsin is secreted in the stomach and acts only under acidic condition and trypsin is secreted in pancreas and acts under alkaline condition.
What is the difference between pepsin and trypsin Brainly?
The main difference between pepsin and trypsin is that the pepsin is secreted by the gastric glands of the stomach whereas the trypsin is secreted by the exocrine glands of the pancreas.. Pepsin and trypsin are two types of proteolytic enzymes secreted by the digestive system in order to digest proteins …..
What are the products of trypsin?
The products of trypsin digestion are amino acids and various polypeptides.
What does Trypsinogen breakdown?
Trypsinogen is a substance that is normally produced in the pancreas and released into the small intestine. Trypsinogen is converted to trypsin. Then it starts the process needed to break down proteins into their building blocks (called amino acids).
What enzyme converts trypsinogen to trypsin?
Enteropeptidase converts trypsinogen into active trypsin, which not only hydrolyses some peptide bonds of food proteins but also activates a number of pancreatic zymogens. For this reason enteropeptidase is a key enzyme in the digestion of dietary proteins and its absence may result in gross protein malabsorption.
What is the function of aminopeptidase Mcq?
Aminopeptidase:- It is the type of exopeptidase. and act on the N-terminal of peptide bond. dipeptidase:- It breaks the dipeptide into each amino acid. It finally converts all the ingested protein into amino acid.
What is aminopeptidase and carboxypeptidase?
Aminopeptidase hydrolyses the peptide bond of the amino acid at the amino terminal of a protein or peptide, releasing a free amino acid. Carboxypeptidase hydrolyses the peptide bond of the amino acid at the carboxyl terminal of a protein or peptide, again releasing a free amino acid.
What are the main functions of HCL and pepsin in digestion?
The HCL present in the gastric juice dissolves bits of food and creates an acidic medium in our stomach. The enzyme pepsinogen is converted to pepsin in this acidic medium. Pepsinogen is a protein-digesting enzyme. It also kills bacteria and other microorganisms that enter along with the food in our stomach.
What are the functions of hydrochloric acid in stomach Mcq?
It creates the acidic conditions necessary for the action of the enzyme pepsin. It activates pepsin to act on proteins. It also destroys the structure of proteins so that the enzymes can digest them easily. It gets mixed with food and kills the bacteria present in food.
What is the function of HCL mucus and pepsin?
Pepsin Pearls Pepsin is a stomach enzyme that serves to digest proteins found in ingested food. Gastric chief cells secrete pepsin as an inactive zymogen called pepsinogen. Parietal cells within the stomach lining secrete hydrochloric acid that lowers the pH of the stomach.
What is the difference between trypsin and trypsin?
Trypsin and chymotrypsin are two very similar digestive enzymes that hydrolyze proteins into amino acids. … This is the main difference between these two enzymes. Activation: The inactive form of trypsin, trypsinogen, is activated by enterokinase, while chymotrypsinogen is activated by trypsin.
What is the substrate of trypsin?
Trypsin is a pancreatic serine protease with substrate specificity based upon positively charged lysine and arginine side chains (Brown and Wold 1973). The enzyme in excreted by the pancreas and takes part in the digestion of food proteins and other biological processes.
What is the work of trypsin tablet?
How does it work ? Trypsin removes dead skin cells (tissue) and allows healthy tissue to grow. Trypsin in combination with other enzymes seems to reduce inflammation and swelling.
What enzymes does trypsin activate?
Trypsinogen is activated by enterokinase, which cleaves an amino-terminal activation peptide (TAP). Active trypsin then cleaves and activates all of the other pancreatic proteases, a phospholipase, and colipase, which is necessary for the physiological action of pancreatic triglyceride lipase.