What is Hypoosmolarity

Ethan Hayes
Published Apr 20, 2026
Hypoosmolarity: In biochemistry, an abnormal decrease in the osmolarity of the body fluids.
What causes Hypoosmolarity?
This can be caused by drinking too many fluids and overhydrating the body. It can also be caused by a condition called hyponatremia, which is a low level of sodium in the blood. Mild cases of hyposmolality usually have no symptoms. Moderate cases can cause nausea and headaches.
What is a Hypoosmolar state?
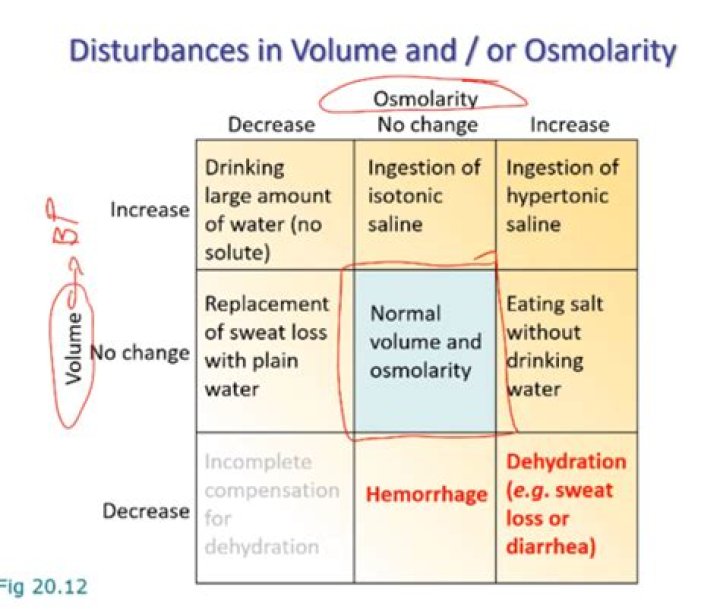
Hypoosmolar hyponatremia is a condition where hyponatremia associated with a low plasma osmolality. The term “hypotonic hyponatremia” is also sometimes used. When the plasma osmolarity is low, the extracellular fluid volume status may be in one of three states: low volume, normal volume, or high volume.
What does osmolarity mean?
The term osmolarity refers to the number of particles of solute per liter of solution, whereas the term osmolality refers to the number of particles of solute per kilogram of solvent.What causes low urine osmolality?
Several conditions may cause low urine osmolality, including: excessive fluid intake, or over-hydration. kidney failure. renal tubular necrosis.
Can HHS lead to DKA?
HHS. The basic underlying mechanism of HHS is a relative reduction in effective circulating insulin with a concomitant rise in counterregulatory hormones. Unlike patients with DKA, most patients with HHS do not develop significant ketoacidosis.
What happens hyponatremia?
In hyponatremia, one or more factors — ranging from an underlying medical condition to drinking too much water — cause the sodium in your body to become diluted. When this happens, your body’s water levels rise, and your cells begin to swell. This swelling can cause many health problems, from mild to life-threatening.
What is urine osmolality test?
An osmolality urine test is performed to measure the concentration of particles in urine. Greater than normal results may indicate conditions such as Addison disease, congestive heart failure or shock.What is osmolarity example?
osmolarity. Osmolarity is dependent upon the number of impermeant molecules in a solution, not on the identity of the molecules. For example, a 1M solution of a nonionizing substance such as glucose is a 1 Osmolar solution; a 1M solution of NaCl = 2 Osm; and a 1M solution of Na2SO4 =3 Osm.
What is tonicity in biology?Tonicity is defined as the ability of a solution surrounding a cell to cause that cell to gain or lose water (Urry et al., 2017). While osmolarity is an absolute quantity, tonicity is relative. … If two solutions have the same concentration of nonpenetrating solutes, they are considered isotonic.
Article first time published onCan hypovolemia cause hyponatremia?
The etiology of hyponatremia is often multifactorial. The most common causes include hypovolemia from gastrointestinal (GI) or other fluid losses, thiazide diuretics, and SIAD [ 1 ].
What's the difference between tonicity and osmolarity?
The key difference between tonicity and osmolarity is that the tonicity measures only the concentration of non-penetrating solutes through a semipermeable membrane while the osmolarity measures the total concentration of penetrating and non-penetrating solutes.
What hormone is secreted during Hypoosmolar state?
In a hypoosmolar state with vasopressin secretion normally suppressed, the kidney can excrete urine with an osmolality as low as 50 mOsm/kg.
What is the normal range for urine osmolality?
An individual with a normal diet and normal fluid intake has a urine osmolality of approximately 500-850 mOsm/kg water. After age 20 years, the upper level of the reference range declines by about 5 mOsm/kg/year.
What is a normal urine sodium level?
For adults, normal urine sodium values are generally 20 mEq/L in a random urine sample and 40 to 220 mEq per day. Your result depends on how much fluid and sodium or salt you take in.
What is normal urine concentration?
Normal Results 1.005 to 1.030 (normal specific gravity) 1.001 after drinking excessive amounts of water. More than 1.030 after avoiding fluids.
What is the most common cause of hyponatremia?
Hyponatremia is decrease in serum sodium concentration < 136 mEq/L (< 136 mmol/L) caused by an excess of water relative to solute. Common causes include diuretic use, diarrhea, heart failure, liver disease, renal disease, and the syndrome of inappropriate antidiuretic hormone secretion (SIADH).
How fast can you correct hyponatremia?
Clinical recommendationEvidence ratingCommentsIn patients with severe symptomatic hyponatremia, the rate of sodium correction should be 6 to 12 mEq per L in the first 24 hours and 18 mEq per L or less in 48 hours.CConsensus guidelines based on systematic reviews
Who is at risk for hyponatremia?
Hyponatremia is more likely in people living with certain diseases, like kidney failure, congestive heart failure, and diseases affecting the lungs, liver or brain. It often occurs with pain after surgery. Also, people taking medications like diuretics and some antidepressants are more at risk for this condition.
Can HHS cause seizures?
In more advanced HHS, presentation is more likely to be altered mental status, seizures and/or coma. Patients may also present with an underlying fever, a clue to an underlying infection.
What is the difference between DKA and HHS?
DKA is characterized by ketoacidosis and hyperglycemia, while HHS usually has more severe hyperglycemia but no ketoacidosis (table 1). Each represents an extreme in the spectrum of hyperglycemia.
What are the symptoms of HHS?
- Blood sugar level of 600 milligrams per deciliter (mg/dL) or 33.3 millimoles per liter (mmol/L) or higher.
- Excessive thirst.
- Dry mouth.
- Increased urination.
- Warm, dry skin.
- Fever.
- Drowsiness, confusion.
- Hallucinations.
What osmolarity is isotonic?
The osmolarity and sodium concentration of isotonic fluids are similar to that of plasma and extracellular fluid. Normal plasma osmolarity is 290 to 310 mOsm/L for dogs and 311 to 322 mOsm/L for cats, and isotonic fluids generally have an osmolality in the range of 270 to 310 mOsm/L.
What is osmolarity of a cell?
Osmolarity is defined as the number of ionic species in molarity that has a characteristic range depending on the species examined. … For example, cell culture media are usually adjusted to 290–320 mOsmol to prevent any osmotic stress to cells.
What is osmolarity in plants?
Osmolarity is a measure of solute concentration, as defined by the number of osmoles of a solute per litre of solution (osmol/L)
What happens if osmolarity is too high?
In healthy people, when osmolality in the blood becomes high, the body releases antidiuretic hormone (ADH). This hormone causes the kidneys to reabsorb water. This results in more concentrated urine. The reabsorbed water dilutes the blood.
What does it mean if your osmolality is high?
“Osmolality” refers to the concentration of dissolved particles of chemicals and minerals — such as sodium and other electrolytes — in your serum. Higher osmolality means you have more particles in your serum. Lower osmolality means the particles are more diluted.
Is high osmolality bad?
Increased or decreased osmolality calls for evaluation of your patient’s fluid and electrolyte balance. The normal range for serum osmolality is 280 to 300 mOsm/kg. Above-normal values may indicate conditions such as dehydration, hyperglycemia, diabetes insipidus, hypernatremia, uremia, and renal tubular necrosis.
What is tonicity in biochemistry?
Tonicity. Tonicity is the capability of a solution to modify the volume of cells by altering their water content. The movement of water into a cell can lead to hypotonicity or hypertonicity when water moves out of the cell.
What is tonicity in physiology?
The ability of an extracellular solution to make water move into or out of a cell by osmosis is known as its tonicity. Tonicity is a bit different from osmolarity because it takes into account both relative solute concentrations and the cell membrane’s permeability to those solutes.
How do you describe tonicity?
Tonicity is the concentration of a solution as compared to another solution. Concentration describes the amount of solutes dissolved by a solution. … In biology, the tonicity of the environment compared to the cell determines how water moves across the semipermeable membrane.