What is electron configuration of elements

William Harris
Published Apr 17, 2026
The electron configuration of an element describes how electrons are distributed in its atomic orbitals. Electron configurations of atoms follow a standard notation in which all electron-containing atomic subshells (with the number of electrons they hold written in superscript) are placed in a sequence.
What is an electron configuration give an example?
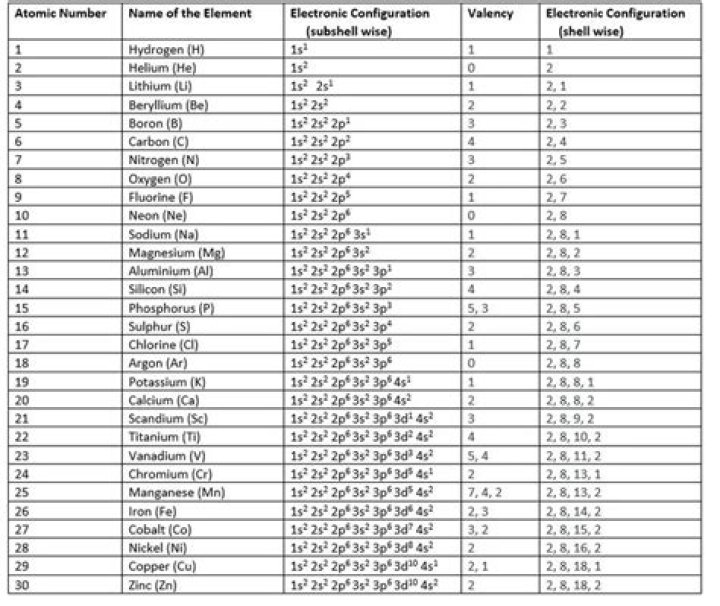
Electron configurations describe where electrons are located around the nucleus of an atom. For example, the electron configuration of lithium, 1s²2s¹, tells us that lithium has two electrons in the 1s subshell and one electron in the 2s subshell.
How do you write the electron configuration?
Writing Electron Configurations. When writing an electron configuration, first write the energy level (the period), then the subshell to be filled and the superscript, which is the number of electrons in that subshell. The total number of electrons is the atomic number, Z.
What is 1s 2s 2p 3s 3p?
In the question 1s 2s 2p 3s 3p represents electron orbital energy levels. … The sequence of orbital energy levels is as always-1s < 2s = 2p < 3s = 3p = 3d <4s = 4p = 4d= 4f. The orbital having the same energy is called a degenerate orbital.What elements have the same electron configuration?
When two different elements have the same electron configuration, they are called isoelectronic.
What is 2p orbital?
p orbitals At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. A p orbital is rather like 2 identical balloons tied together at the nucleus.
How do you calculate configuration?
To calculate an electron configuration, divide the periodic table into sections to represent the atomic orbitals, the regions where electrons are contained. Groups one and two are the s-block, three through 12 represent the d-block, 13 to 18 are the p-block and the two rows at the bottom are the f-block.
What is electron configuration used for?
The electron configuration is used to describe the orbitals of an atom in its ground state, but it can also be used to represent an atom that has ionized into a cation or anion by compensating with the loss of or gain of electrons in their subsequent orbitals.What is the next atomic orbital in the series 1s2s2p3s3p4s?
This is whole series. In this order electron get accommodate in various orbitals or subshells. The next atomic orbitals in the series are : 1s,2s,2p,3s,3p,4s,3d,4p,5s,4d,5p,6s,4f,5d,6p,7s …..
Why do different elements have different electron configurations?Many of the physical and chemical properties of elements can be correlated to their unique electron configurations. The valence electrons, electrons in the outermost shell, are the determining factor for the unique chemistry of the element.
Article first time published onWhat is e9 class configuration?
Electronic configuration is defined as the distribution of electrons into the orbitals of an atom. Every neutral atom consists of a fixed number of electrons which is equal to the number of protons and is called the atomic number.
How do you find the electronic configuration of an element in Class 9?
The electron configuration of an atom is written with the help of subshell labels. These labels contain the shell number (given by the principal quantum number), the subshell name (given by the azimuthal quantum number) and the total number of electrons in the subshell in superscript.
What is the shape of DXY?
The ml values for five d orbitals are -2, -1, 0, +1, and +2 i.e., we can say d-subshell has five orientations. All these d-orbitals have the same energy and are called degenerate orbitals. The shape of the d-orbitals is given below: Hence, we can say d-orbitals have double dumbbell-shaped.
What is shape of P orbital?
A p orbital has the approximate shape of a pair of lobes on opposite sides of the nucleus, or a somewhat dumbbell shape. An electron in a p orbital has equal probability of being in either half.
What is 1s orbital?
The closest orbital to the nucleus, called the 1s orbital, can hold up to two electrons. This orbital is equivalent to the innermost electron shell of the Bohr model of the atom. It is called the 1s orbital because it is spherical around the nucleus. The 1s orbital is always filled before any other orbital.
What comes after 5s in electron configuration?
The 4d sublevel is filled next, after the 5s sublevel.
What does the Aufbau principle suggest about electron configuration?
The Aufbau principle states that electrons fill lower-energy atomic orbitals before filling higher-energy ones (Aufbau is German for “building-up”). By following this rule, we can predict the electron configurations for atoms or ions.
How is the size and shape of an electron cloud determined?
The answer to this is that the shape of an orbital is dependent upon how many electrons can be held within the orbital. We already know that s-orbitals hold two electrons. The shape of this orbital is a sphere.
What is the electronic configuration of 15p?
Therefore the Phosphorus electron configuration will be 1s22s22p63s23p3. The configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom. This makes it easier to understand and predict how atoms will interact to form chemical bonds.
What is the correct configuration of p15?
Group1544.15°C, 111.47°F, 317.3 KBlockp1.823 (white)Atomic number1530.974State at 20°CSolid31PElectron configuration[Ne] 3s23p37723-14-0
What is the electron configuration for the atom with 15 electrons?
ElementAtomic numberElectron configurationaluminum131s22s22p63s23p1silicon141s22s22p63s23p2phosphorus151s22s22p63s23p3sulfur161s22s22p63s23p4
What is the element 1s2 2s2 2p6?
ABFluorine1s2 2s2 2p5Neon1s2 2s2 2p6Sodium1s2 2s2 2p6 3s1Magnesium1s2 2s2 2p6 3s2
What is the electronic configuration of 13al?
NameAluminiumBlock in Periodic Tablep-blockElectronic Configuration[Ne] 3s2 3p1Melting Point660.32 °CBoiling Point2519 °C
Which element has the following electron configuration 1s22s22p63s23p64s23d8?
Answer and Explanation: The atomic number is 28 which is of nickel (Ni).
When writing an electron configuration first write the?
How to Write an Electron Configuration. The symbols used for writing the electron configuration start with the shell number (n) followed by the type of orbital and finally the superscript indicates how many electrons are in the orbital. For example: Looking at the periodic table, you can see that Oxygen has 8 electrons …