What is an example of a dehydration synthesis

Olivia Zamora
Published Apr 20, 2026
Answer Not Found
What are three examples of dehydration synthesis reactions?
- Alcohol. Alcohols undergo dehydration reaction in the presence of a strong acid like sulfuric acid or phosphoric acid to yield alkenes. …
- Carbohydrate. In the dehydration synthesis of carbohydrates like sugar, two monosaccharides combine to form a disaccharide. …
- Amino acid.
What is an example of dehydration synthesis in cellular respiration?
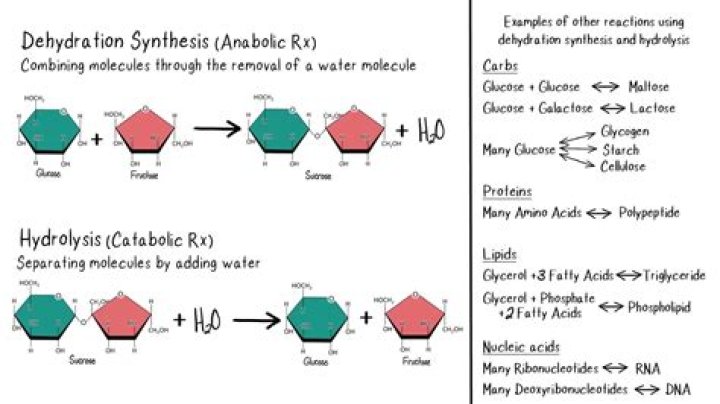
Many different biological or chemical processes utilize the dehydration synthesis reaction. Examples include the formation of disaccharides, like sucrose, from monosaccharides, like glucose, and alcohol condensation to make an ether product. An acid catalyst can be used during a dehydration synthesis reaction.
What is an example of dehydration synthesis quizlet?
a chemical reaction in which two or more molecules bond by losing one or more water molecules. Plants build starches through dehydration synthesis. … Examples of disaccharides include maltose (two glucose molecules) and sucrose (one glucose molecule and one fructose molecule).What is dehydration give example?
A dehydration reaction is a chemical reaction between two compounds where one of the products is water. For example, two monomers may react where a hydrogen (H) from one monomer binds to a hydroxyl group (OH) from the other monomer to form a dimer and a water molecule (H2O).
Is photosynthesis a dehydration synthesis?
Glucose molecules are combined by dehydration synthesis to form cellulose, which is used as a structural material. Dehydration synthesis is also used to convert glucose to starch, which plants use to store energy.
What is dehydration synthesis kid definition?
Dehydration synthesis is the creation of larger molecules from smaller monomers where a water molecule is released. This can be used in the creation of synthetic polymers such as polyethylene terephthalate (PET), or the creation of large biological molecules such as carbohydrate polymers and triglycerides.
What occurs during a dehydration synthesis?
Dehydration synthesis involves the formation of new chemical bonds between two molecules which leads to the formation of new compounds. A reaction occurs with the loss of water molecule at each step.What does dehydration synthesis do quizlet?
What is dehydration synthesis? The process of bonding monomers by taking out the OH from one and taking out the H from the other. This frees up a spot for the molecules from which the OH and H were taken to bond. The H and OH bond to make a water molecule.
What does dehydration synthesis produce quizlet?Dehydration synthesis is involved in reactions which combine organic monomers to produce polymers. For each covalent bond produced, a water molecule is created. d. Dehydration synthesis reactions are high energy reactions that do not require enzymes.
Article first time published onIs a dehydration synthesis reaction An example of a catabolic or anabolic reaction?
Dehydration synthesis is an anabolic reaction.
What is dehydration synthesis and hydrolysis?
Dehydration synthesis reactions build molecules up and generally require energy, while hydrolysis reactions break molecules down and generally release energy. Carbohydrates, proteins, and nucleic acids are built up and broken down via these types of reactions, although the monomers involved are different in each case.
Is dehydration synthesis Exergonic or Endergonic?
A dehydration synthesis is an endergonic (or ‘energy in’) type of reaction that cannot take place without the input of energy from somewhere else.
What is the dehydration synthesis equation?
Dehydration reactions RCO2H + R′OH ⇌ RCO2R′ + H2O. Two monosaccharides, such as glucose and fructose, can be joined together (to form saccharose) using dehydration synthesis. The new molecule, consisting of two monosaccharides, is called a disaccharide.
Why is it called dehydration synthesis?
This type of reaction is known as dehydration synthesis, which means “to put together while losing water. ” It is also considered to be a condensation reaction since two molecules are condensed into one larger molecule with the loss of a smaller molecule (the water.) …
Which one of the following is an example of dehydrating agent used for desiccation?
Some examples include silica gel, bauxite, anhydrous Calcium chloride and montmorillonite clay. Desiccants also find use in industrial applications, like humidity detection, waste removal and cleaning, and also regenerative drying.
Where does dehydration synthesis occur in the body?
In biological systems, dehydration synthesis reactions occur in every cell, especially since it is important for the formation of ATP. Nearly all biopolymers are also derived from this reaction.
What is a dehydration reaction in biology?
In biology and chemistry, a dehydration synthesis reaction (or a condensation reaction) is one that synthesizes—or joins—two molecules together, resulting in a loss of water.It may not sound like much, but the underlying importance of dehydration synthesis is that it is central to the production of larger biological …
Do plants use dehydration synthesis?
In most autotrophic organisms utilize dehydration synthesis in order to form long chains of small molecules. For instance, this process used for storing excess glucose by converting it to glycogen (storage form of carbohydrates in animals) or starch (storage form of carbohydrates) in plants.
Is amylose formed by dehydration synthesis?
Polysaccharides are formed when a dehydration reaction occurs between a large number of monosaccharides. Amylose (below) is a polysaccharide composed of between 60 and 300 glucose units. The difference in orientation of the glucose molecules is why humans are incapable of digesting cellulose. …
Is water a reactant or product in dehydration synthesis?
Dehydration Synthesis The monomers combine with each other using covalent bonds to form larger molecules known as polymers. In doing so, monomers release water molecules as byproducts. This type of reaction is known as dehydration synthesis, which means “to put together while losing water.”
What is dehydration synthesis and hydrolysis quizlet?
Hydrolysis. A process where a molecule splits into two or more smaller molecules and gains the “parts” of water on the broken ends. Dehydration synthesis. A process where two molecules lose “parts” of water and join/bond together.
What is dehydration synthesis and why is it called this quizlet?
complex carbohydrates, nucleic acids, and proteins. what does dehydration synthesis mean and what is it also called and way. to put together while losing water and its also known as condensation reaction since two molecules are condensed into one larger molecule with the loss of a smaller molecule (the water)
What is dehydration biology quizlet?
Dehydration Reaction. A chemical reaction in which two molecules covalently bond to each other with the removal of water. Denaturation. In protiens, a process in which a protien unravels and loses its native conformation, thereby becoming biologically inactive.
What are cellulose and starch are examples of?
Starch, glycogen, cellulose, and chitin are primary examples of polysaccharides. Starch is the stored form of sugars in plants and is made up of a mixture of amylose and amylopectin (both polymers of glucose).
What do hydrolysis and dehydration synthesis have in common?
Dehydration and hydrolysis reactions are similar for all macromolecules, but each monomer and polymer reaction is specific to its class. Dehydration reactions typically require an investment of energy for new bond formation, while hydrolysis reactions typically release energy by breaking bonds.
What macromolecules use dehydration synthesis?
Complex carbohydrates, nucleic acids, and proteins are all examples of polymers that are formed by dehydration synthesis. Monomers like glucose can join together in different ways and produce a variety of polymers.
Is dehydration synthesis endothermic or exothermic?
Typically, all Dehydration Synthesis reactions, where smaller compounds are bonded together to produce larger compounds, are endothermic.
Which of the following bond formation is dehydration synthesis?
(D) Peptide bond, glycosidic bond and phosphodiester bond formation is dehydration synthesis.
Is ATP used in dehydration synthesis?
ATP captures small quantities of energy by binding one phosphate unit to adenosine diphosphate (ADP) to synthesize, by dehydration synthesis, a high-energy molecule of ATP. … ATP can be produced in catabolic reactions in several ways.
Are dehydration reactions exergonic?
Dehydration synthesis, that is, builds molecules up – at the expense of energy (endergonic reaction) – while hydrolysis breaks molecules apart, liberating energy (exergonic reaction).