What can shift equilibrium

Olivia Zamora
Published Mar 28, 2026
According to Le Chatelier’s principle, adding additional reactant to a system will shift the equilibrium to the right, towards the side of the products. … If we add additional product to a system, the equilibrium will shift to the left, in order to produce more reactants.
What causes equilibrium shifts?
If the stress applied to a system in equilibrium is a change in the concentration of a component of the equilibrium, the system shifts to counteract that change. If the concentration of a substance is increased, the reaction that consumes that substance is favored, and the equilibrium shifts away from that substance.
What four factors can shift the position of an equilibrium?
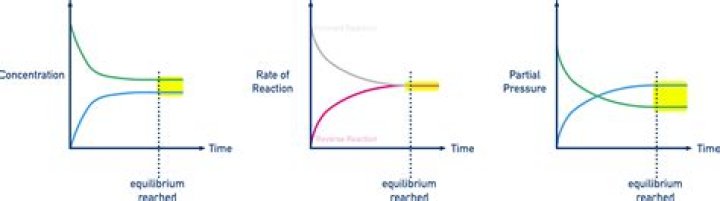
Changes in concentration, temperature, and pressure can affect the position of equilibrium of a reversible reaction. Chemical reactions are equilibrium reactions. Equilibrium occurs when a certain proportion of a mixture exists as reactants and the rest exits as products.
What are the 3 stresses that affect equilibrium?
The three most common ways to stress a system at equilibrium are changing the concentration of one of the reactants or products, changing the temperature of the system, or changing the pressure on the system.Which change will shift the equilibrium to the right?
For an exothermic reaction, heat is a product. Therefore, increasing the temperature will shift the equilibrium to the left, while decreasing the temperature will shift the equilibrium to the right.
What is equilibrium position in physics?
When all the forces that act upon an object are balanced, then the object is said to be in a state of equilibrium. … If an object is at equilibrium, then the forces are balanced. Balanced is the key word that is used to describe equilibrium situations. Thus, the net force is zero and the acceleration is 0 m/s/s.
What is equilibrium position in chemistry?
the equilibrium position: The point in a chemical reaction at which the concentrations of reactants and products are no longer changing.
What factors do not affect equilibrium?
Effect of a Catalyst There is no effect on the equilibrium composition of a reaction mixture. This is because catalyst increases the speed of both forward and backward reactions to the same extent in a reversible reaction.What stress can result in a shift in equilibrium during a reaction?
If a stress is applied by reducing the concentration of a product, the equilibrium position will shift toward the products.
What stress would cause the equilibrium to shift to the left?If a stress is applied by increasing the concentration of a product, the equilibrium position will shift toward the left, reducing the concentration of the product.
Article first time published onWhat factors change the equilibrium constant?
The only thing that changes an equilibrium constant is a change of temperature. The position of equilibrium is changed if you change the concentration of something present in the mixture.
What factors affect equilibrium price?
Overview of Changes in Equilibrium Prices. As you can see, an increase in demand causes the equilibrium price to rise. On the other hand, a decrease in demand causes the equilibrium price to fall. An increase in supply causes the equilibrium price to fall, while a decrease in supply causes the equilibrium price to rise …
Which change would not cause a shift in the equilibrium?
Answers. Because there is an equal number of moles on both sides of the reaction, an increase in volume will have no effect on the equilibrium and thus there is no shift in the direction. Similarly, when you decrease the volume there is no effect on the equilibrium.
How does Catalyst affect equilibrium?
Adding a catalyst to a reaction at equilibrium has no effect on the position of equilibrium. It does however allow equilibrium to be reached more quickly, or established at a lower temperature, which makes reactions more profitable.
What is equilibrium position in economics?
Economic equilibrium is a condition or state in which economic forces are balanced. … The point of equilibrium represents a theoretical state of rest where all economic transactions that “should” occur, given the initial state of all relevant economic variables, have taken place.
What is equilibrium position in waves?
The equilibrium is represented by the horizontal line in the middle of the wave. One period is the time it takes to go for one cycle, which means to get from one crest to crest, trough to trough, or to and from corresponding equilibrium points (both equilibrium points same direction).
What does the equilibrium position depend on?
The equilibrium constant is independent of the initial concentrations of reactants. The factors that affect the value of the equilibrium constant are temperature, nature of the solvent, the ionic strength of ions in the reaction mixture, etc.
Can an object in equilibrium be moving?
A moving object is in equilibrium if it moves with a constant velocity; then its acceleration is zero. A zero acceleration is the fundamental characteristic of an object in equilibrium.
What are the 3 types of equilibrium?
There are three types of equilibrium: stable, unstable, and neutral. Figures throughout this module illustrate various examples.
What is equilibrium position in simple harmonic motion?
At the equilibrium position, the velocity is at its maximum and the acceleration (a) has fallen to zero. Simple harmonic motion is characterized by this changing acceleration that always is directed toward the equilibrium position and is proportional to the displacement from the equilibrium position.
How can equilibrium state be disturbed?
Systems at equilibrium can be disturbed by changes to temperature, concentration, and, in some cases, volume and pressure; volume and pressure changes will disturb equilibrium if the number of moles of gas is different on the reactant and product sides of the reaction.
What happens when equilibrium is disturbed?
When a chemical system at equilibrium is disturbed, it returns to equilibrium by counteracting the disturbance. As described in the previous paragraph, the disturbance causes a change in Q; the reaction will shift to re-establish Q=K.
What Stress affects the value of an equilibrium constant?
If a system at equilibrium is subjected to a perturbance or stress (such as a change in concentration) the position of equilibrium changes. Since this stress affects the concentrations of the reactants and the products, the value of Q will no longer equal the value of K.
How does pressure affect equilibrium?
When there is an increase in pressure, the equilibrium will shift towards the side of the reaction with fewer moles of gas. When there is a decrease in pressure, the equilibrium will shift towards the side of the reaction with more moles of gas.
What would happen to the position of the equilibrium when the following changes are made to the equilibrium system below 2SO3 2SO2 O2?
2) What would happen to the position of the equilibrium when the following changes are made to the equilibrium system below? 2SO3(g) ↔ 2SO2(g) + O2(g) (a) Sulfur dioxide is added to the system. Equilibrium will shift to favor reactants.
What factor is equilibrium constant independent?
Characteristics of Equilibrium Constant: It is independent of the initial concentrations of the reacting species. It changes with the change in the temperature. It depends on the nature of the reaction. It is independent of the change of pressure, volume and concentrations of the reactants and products.
What is the most important factor that affects chemical equilibrium?
It states that changes in the temperature, pressure, volume, or concentration of a system will result in predictable and opposing changes in the system in order to achieve a new equilibrium state.
What are the five factors that shift supply?
There are a number of factors that cause a shift in the supply curve: input prices, number of sellers, technology, natural and social factors, and expectations.
What causes equilibrium price to fall?
A decrease in demand will cause the equilibrium price to fall; quantity supplied will decrease. An increase in supply, all other things unchanged, will cause the equilibrium price to fall; quantity demanded will increase. A decrease in supply will cause the equilibrium price to rise; quantity demanded will decrease.
What change is illustrated by the shift taking place on this graph?
An increase in supply is illustrated by the shift taking place on this graph.
Does increasing pressure shift equilibrium?
If the pressure is increased, the position of equilibrium moves in the direction of the fewest moles of gas. … Therefore, if the pressure is increased, the position of equilibrium will move to the right and more methanol will be produced.