What are isotonic crystalloid fluids

Ava Wright
Published Apr 15, 2026
Isotonic fluids have a similar sodium concentration as the extracellular fluid compartment and have minimal impact on intracellular volume. Isotonic crystalloid fluids can vary in their concentration of the electrolytes sodium, chloride, potassium, magnesium, and calcium (Table 1).
What is an example of an isotonic crystalloid solution?
Isotonic sodium chloride solution (normal saline [NS]) and lactated Ringer (LR) solution are isotonic crystalloid fluids, the standard intravenous (IV) fluids used for initial volume resuscitation.
What are examples of crystalloid fluids?
- Lactated Ringer’s/Hartman’s solution (lactate buffered solution)
- Acetate buffered solution.
- Acetate and lactate buffered solution.
- Acetate and gluconate buffered solution.
- 0.45% NaCl (hypotonic solution)
- 3% NaCl (hypertonic solution)
- 5% Dextrose in water.
What are the three types of Crystalloids?
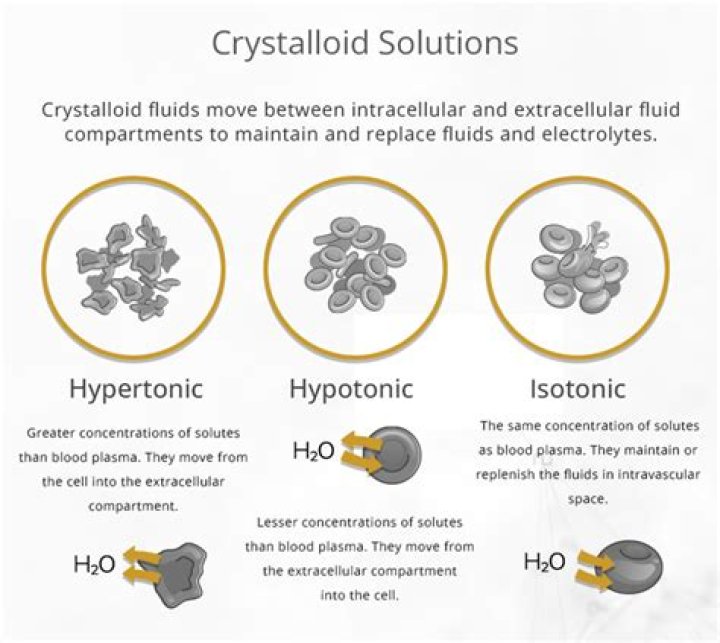
Types of Crystalloid Solutions There are three tonic states: isotonic, hypertonic, and hypotonic.What are isotonic fluids?
Isotonic solutions are IV fluids that have a similar concentration of dissolved particles as blood. An example of an isotonic IV solution is 0.9% Normal Saline (0.9% NaCl). … Isotonic solutions are used for patients with fluid volume deficit (also called hypovolemia) to raise their blood pressure.
What are crystalloid fluids?
Crystalloid fluids are a subset of intravenous solutions that are frequently used in the clinical setting. Crystalloid fluids are the first choice for fluid resuscitation in the presence of hypovolemia, hemorrhage, sepsis, and dehydration.
What is isotonic solution example?
A solution is isotonic when its effective mole concentration is the same as that of another solution. This state provides the free movement of water across the membrane without changing the concentration of solutes on either side. Some examples of isotonic solutions are 0.9% normal saline and lactated ringers.
Is dextrose a crystalloid?
Crystalloids are the most common fluids used in the healthcare setting. The following are some examples of the most common solutions in the crystalloid category. Dextrose 5% is consists of 278 mmoL/L of dextrose. The pH is 4.0 and the osmolarity is around 272.Is lactated Ringer's a crystalloid or colloid?
Crystalloids, such as saline and Ringer’s lactate, are solutions of salt, water and minerals, and are commonly used in the clinical setting. They have small molecules, and, when used intravenously, they are effective as volume expanders.
Is saline isotonic?0.9% saline is a perfectly isotonic solution that is isoosmolar to human plasma and is not associated with hypernatremia [2].
Article first time published onIs LRS an isotonic crystalloid?
LRS has the lowest osmolarity and lowest sodium concentration (130 meq/L) compared to other isotonic crystalloids. The only major contraindication for use of LRS is in-line (i.e., via the same catheter) administration with blood products.
How fast is isotonic crystalloid?
Current recommendations are to administer 20 mL/kg of fluid as a bolus over 5 to 10 minutes and repeat as needed.
What is the difference between colloids and Crystalloids?
The main difference between colloid and crystalloid is their particle size. Colloidal systems have much larger particles compared to crystalloid systems. Hence, the permeability of colloidal systems is lower than that of crystalloid systems.
What is isotonic hypertonic solution?
Isotonic solutions. Isotonic solutions are two solutions that have the same concentration of a solute. Hypertonic solution. Hypertonic solution is one of two solutions that has a higher concentration of a solute.
What are isotonic hypertonic and hypotonic solutions?
The mixture of a solute in a solvent is called a solution. … In comparing two solutions of unequal solute concentration, the solution with the higher solute concentration is hypertonic, and the solution with the lower solute concentration is hypotonic. Solutions of equal solute concentration are isotonic.
Is distilled water isotonic?
NaCl is isotonic to the red blood cell at a concentration of 154 mM. This corresponds with NaCl 0.9%. … Distilled water on the other hand is hypotonic to red blood cells.
Which is same in isotonic solution?
An isotonic solution is one that has the same osmolarity, or solute concentration, as another solution. If these two solutions are separated by a semipermeable membrane, water will flow in equal parts out of each solution and into the other.
Why is isotonic solution used?
Isotonic fluids Isotonic solutions are used: to increase the EXTRACELLULAR fluid volume due to blood loss, surgery, dehydration, fluid loss that has been loss extracellularly.
Is d10 isotonic?
Dextrose 10% in Water (D10W) is an hypertonic IV solution used in the treatment of ketosis of starvation and provides calories (380 kcal/L), free water, and no electrolytes.
Is dextrose colloid or crystalloid?
Crystalloids are the most common fluids used in the healthcare setting. The following are some examples of the most common solutions in the crystalloid category. Dextrose 5% is consists of 278 mmoL/L of dextrose. The pH is 4.0 and the osmolarity is around 272.
When do you not use LR?
- Liver disease.
- Lactic acidosis, which is when there is too much lactic acid in your system.
- A pH level greater than 7.5.
- Kidney failure.
Is sodium bicarbonate a crystalloid?
The use of hypertonic crystalloid solutions, including sodium chloride and bicarbonate, for treating severe sepsis has been much debated in previous investigations.
Is FFP a colloid?
Colloids have larger molecules and may be more efficient at increasing fluid volume in the blood. They include starches, dextrans, gelatins, and naturally-occurring colloids, such as albumin or fresh frozen plasma or FFP.
What are lactated ringers?
The contents of Ringer’s lactate include sodium, chloride, potassium, calcium, and lactate in the form of sodium lactate, mixed into a solution with an osmolarity of 273 mOsm/L and pH of about 6.5. In comparison, normal saline (NS) has an osmolarity of about 286 mOsm/L.
Is glucose a crystalloid?
Crystalloid intravenous fluids, which include solutions containing small molecular weight solutes such as sodium, chloride and glucose, are the most common type of fluid used to replace blood in the United States.
Is .45 NaCl isotonic?
Sodium Chloride 0.45% Solution for Infusion is a hypotonic solution, with an approximate osmolarity of 154 mOsm/l. The pharmacodynamic properties of the solution are those of the sodium and chloride ions in maintaining the fluid and electrolyte balance.
What is isotonic to plasma?
Isotonic solutions contain an electrolyte balance similar to plasma in the bloodstream. When an isotonic solution is administered, the fluid volume of the patient is increased without a fluid shift. Common examples of isotonic solutions are 0.9% normal saline and lactated ringers.
Can I Nebulize with saline?
How to use Saline 0.9 % Solution For Nebulization. This medication is used with a special machine called a nebulizer that changes the solution to a fine mist that you inhale. Learn how to prepare the solution and use the nebulizer properly.
Is D5W isotonic?
D5W is unique. It can be categorized as an isotonic solution or a hypotonic solution. The amount of dextrose in the solution makes its tonicity, or solute concentration, similar to that of intravascular fluid, making it isotonic.
What is d5 saline?
Dextrose and Sodium Chloride Injection, USP (dextrose and sodium chloride inj) is a sterile, nonpyrogenic solution for fluid and electrolyte replenishment and caloric supply in single dose containers for intravenous administration. It contains no antimicrobial agents.
What fluids do you give for sepsis?
Answer: Crystalloid solutions remain the resuscitative fluid of choice for patients with sepsis and septic shock. Balanced crystalloid solutions may improve patient-centered outcomes and should be considered as an alternative to 0.9% normal saline (when available) in patients with sepsis.