Is Acetanilide soluble in methanol

William Harris
Published Apr 23, 2026
The solubility of acetanilide is 18.5 g in 100 ml of methanol at 0 o C, and 59.2 g in 100 ml of methanol at 60 o C.
Does acetanilide dissolve in ethanol?
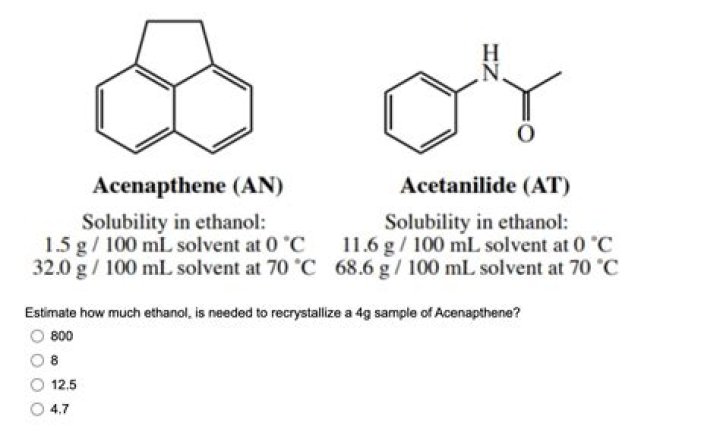
The solubility values of acenapthene and acetanilide in ethanol are shown in Figure 3.28. Since acetanilide (AT) is more soluble than acenapthene at this temperature (see Figure 3.28), it should also completely dissolve in 12.5mL of hot ethanol. … The acetanilide impurity, however, should not crystallize.
What is methanol soluble in?
Methanol is soluble in water. Like water, methanol has relatively strong hydrogen bonding acting between its molecules. The attraction between methanol and water is strong enough to overcome the methanol-methanol and water-water intermolecular forces of attraction.
What is the best solvent for acetanilide?
Water is the best solvent to crystallize acetanilide because water is the cheapest solvent and its solubility in water at 100°C is ten times its solubility at 25°C. So dissolve acetanilide in boiling water and cool the solution to room temperature when acetanilide crystallizes out.Why is ethanol not a good solvent for recrystallization acetanilide?
I would expect acetanilide to be readily soluble in ethyl acetate at room temperature, making it unsuitable for this recrystallization. It’s not enough to be soluble. You want a solvent to have high solubility at high temperature, plus you want to have low solubility of the solute at low temperature.
What makes acetanilide soluble in water?
The reason for why water is able to dissolve a number is substances, is due to this polarity that enables water to separate polar solute molecules. Waters polarity makes gives it the property of being a good solvent. … When the water was boiled at a temperature below the acetanilide melting point.
Is acetanilide soluble in benzene?
Acetanilide Physical and Chemical Properties Solubility: Acetanilide is a little soluble in water. It is also soluble in diethyl ether, ethanol, benzene and acetone.
Is acetanilide soluble in hot water?
Acetanilide has a much higher solubility in hot water than in cold water. … A successful recrystallization requires that the compound be soluble at the solvent’s boiling point and insoluble at low temperature.Is acetanilide soluble in HCl?
Explanation: aniline, the nitrogen atom donates its lone pair to the acid and by forming a salt (ionic compound), it dissolves in water(aqueous HCl). … That’s why acetanilide is not soluble in aqueous HCl.
Why is methanol infinitely soluble?Solubility of methanol in water is due to attraction between partially negative alcoholic oxygen of methanol with partial positive Hydrogen of water molecule. This association results in the formation of Inter molecular Hydrogen bonding.
Article first time published onWhy is methanol more soluble than methane?
Methanol is more soluble in water because it is more polar than methane. Methane is non-polar while water is polar.
Why are methanol and water soluble in each other?
The solubility of lower alcohols in water is due to the formation of a hydrogen bond between alcohols and the water molecules. … Therefore, the methanol is soluble in water due to hydrogen bonding between methanol and water molecules.
What solvent is used in the recrystallization of acetanilide Why?
Crude acetanilide looks like grains of brown rice, while pure acetanilide forms shiny crystals in cold water. The crude solid is dissolved in the smallest possible amount of solvent of choice; in this case the solvent is water. Acetanilide has a much higher solubility in hot water than in cold water.
Which solvent is suitable for recrystallization of acetanilide?
Why is water a good solvent for the recrystallization of acetanilide? (Refer to a standard chemistry handbook for the solubility of acetanilide in hot and cold water.)
Is acetanilide soluble in acetic acid?
You can imagine half of the acetic anhydride sticking to the nitrogen atom on the amine, while the other half takes off with one of the hydrogen atoms. The acetic acid product is, of course, quite soluble in water, while the acetanilide product is not very soluble and falls out of solution as a precipitate.
Is acetanilide soluble in nahco3?
Acetanilide and phenacetin are both organic compounds that readily dissolve into dichloromethane. As an organic, non charged compound, they will not dissolve readily into an aqueous solution, such as that of sodium bicarbonate.
How is acetanilide metabolized?
The route of metabolism of acetanilide in man was shown to be as follows: a minor fraction of the drug deacetylates to form aniline; this compound was shown to be the precursor of the substance which oxidizes hemoglobin to methemoglobin; the major fraction of the drug is oxidized to N-acetyl p-aminophenol; this …
Does acetanilide dissolve in hexane?
Firstly you could dissolve your acetanilide in hot ethanol and add hexane dropwise (keeping the solution hot) until you detect some cloudiness then leave it to cool. Secondly suspend your acetanilide in hot hexane and add ethanol dropwise (keeping the solution hot) until it dissolves then leave it to cool.
Is a derivative of acetanilide?
Para red is considered a derivative of acetanilide, simply because it’s ‘derived’ from the compound as a building block. Another important derivative of acetanilide is a molecule called sulfanilamide.
Is acetanilide soluble in chloroform?
Soluble in hot water, ethanol, ether, chloroform, acetone, glycerol and benzene.
Is acetanilide a base?
ACETANILIDE is an amide. … Amides are very weak bases (weaker than water). Imides are less basic yet and in fact react with strong bases to form salts. That is, they can react as acids.
Is acetanilide more polar than water?
Water was chosen as the recrystallization solvent for this particular recrystallization. Water is slightly more polar than acetanilide. Both have sights for hydrogen bonding, but the geometry of the water molecule causes it to be slightly more polar than the amide group on acetanilide.
What happens when acetanilide dissolves in hot water?
Acetanilide has a much higher solubility in hot water than in cold water. The purified solid will not recrystallize later in the experiment if too much hot solvent is added in the beginning. … Any soluble impurities remain in solution during the cold filtration, while the purified solid remains on the filter paper.
Why is Acetanilide less soluble than aniline?
To the amino group of -NH2 an acetyl group is attached, which is electron attracting ie., it has a -I effect. So this makes the availability of the lone pair of electron on the Nitrogen Atom of the Amino group in acetanilide less. It makes the anilide less basic than ANILINE.
Why aniline is soluble in HCl?
Aniline is more soluble in aq. … Aniline does not undergo hydrogen bonding because of the presence of the benzene which is highly hydrophobic. therefore aniline is insoluble in water. In the HCl the amine group becomes protonated (-NH3+) and the ionic hence soluble in HCl.
Why aniline is soluble in dilute hydrochloric acid?
Aniline dissolves in hydrochloric acid because of the basic nature of aniline it forms salt with HCl. … (ii) If the solution is turbid or ppt appears and remains unaffected by the addition of an acid, the given amine is a secondary amine.
What product is formed in the bromination of acetanilide?
Bromination of acetanilide gives para brominated acetanilide mainly, because amino group of acetanilide is protected by acetyl group. Reaction: Mechanism: Bromination is an electrophilic substitution reaction on an aromatic ring.
Does methanol dissolve in methanol?
NameFormulaSolubilityMethanolCH3OHmiscibleEthanolC2H5OHmisciblePropanolC3H7OHmiscible
Which is more soluble methanol or ethanol?
For alcohols, it is known that as the number of carbon chain or skeleton increases the lesser its solubility in that polar solvent. Ethanol has one extra carbon in its skeleton than methanol. Hence, methanol will be more soluble in water than ethanol. Methanol is more soluble.
Why is methanol insoluble in hexane?
The general rule is that ‘polar molecules dissolves in polar solvents and non-polar solvents dissolves in non-polar solvents’, that is, “like dissolves in like”. … Hexane is a nonpolar solvent; the interaction between polar methanol and is relatively weak. Therefore, the solubility of methanol in hexane is very less.
Which compound is most soluble in methanol?
CompoundCH3CH2OHIUPAC NameEthanolCommon NameEthyl alcoholMelting Poing (oC)-114.7Boiling Point (oC)78.5