Indicator chemistry - How To Discuss

David Jones
Published Apr 26, 2026
Indicator chemistry
What does an indicator do in chemistry? Indicator (chemical composition). A substance that changes color to indicate the presence of an ion or substance can be used to indicate the completion of a chemical reaction or (in medicine) to test for a specific reaction.
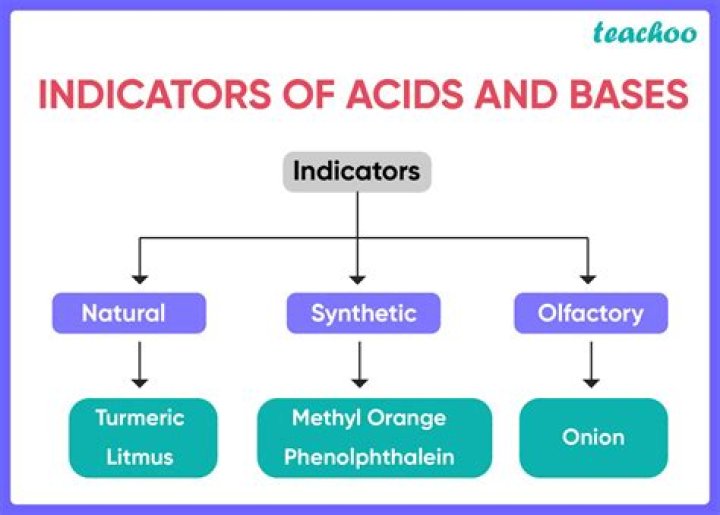
What are some examples of indicators in chemistry?
- Methyl Violet
- Azolythmine
- Leuco-malachite green
- Methyl red
- Thymol blue
- Bromocresol Green
- methyl yellow
- orange brandy
- Bromophenol blue
- Congo red
How is an indicator defined in chemistry?
Indicators are substances whose solutions change color due to changes in pH. They are known as acid-base indicators. They are usually weak acids or bases, but their conjugate bases or acid forms have different colors due to different absorption spectra.
What are indicators used for chemistry?
What is the definition of indicator in chemistry? Indicators are substances that change color when they come into contact with acids and bases. The most commonly used indicator in the laboratory is sunflower. It is made from lichen and comes as a solution or as a strip of paper called litmus paper.
How do indicators work in chemistry?
Indicators are large organic molecules used in chemistry to determine the pH of a substance. They take on different colors depending on whether they are added to an acid, a base (also called an alkali), or a neutral substance. Most indicators themselves are weak acids and respond to changes in the concentration of hydrogen ions.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What is the purpose of an indicator in chemistry quizlet
What is the purpose of an indicator in chemistry quizlet
Chemical indicators are especially useful in chemistry to determine when a reaction has undergone a chemical change or is complete.
What are the indicators that are used in chemistry?
Sunflower, phenolphthalein and methyl orange are commonly used indicators in the laboratory. The sunflower indicator solution turns red in acidic solutions, blue in alkaline solutions, and violet in neutral solutions.
What are some examples of chemical indicators?
A chemical indicator, any substance that gives a visible indication, usually by a color change, of the presence or absence of a threshold concentration of a chemical such as an acid or an alkali in solution. An example is a substance called methyl yellow, which gives an alkaline solution a yellow color.
What are some examples of indicators in chemistry definition
Some examples of indicators are sunflower, turmeric, phenolphthalein, etc. called a chemical indicator.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What is a Class 1 chemical indicator?
What is a Class 1 chemical indicator?
Class I Indicator Class I indicators respond to one or more critical process variables. An example of a Class I chemical indicator is indicator tape that is attached to the outside of a package and is primarily used to secure packaged instrument cassettes. After removing the plastic/paper wrapper or wrapped cassette.
What are some examples of indicators in chemistry examples
Some examples of indicators are sunflower, turmeric, phenolphthalein, etc. called a chemical indicator.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What are the uses of a chemical indicator?
What are the uses of a chemical indicator?
Chemical indicators are used as internal and external indicators and as part of standard performance and stress tests. It is important to note that chemical indicators alone do not confirm the sterility of an item.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() How is an indicator defined in chemistry made
How is an indicator defined in chemistry made
Indicators are substances that change color when they come into contact with acids and bases. The most commonly used indicator in the laboratory is sunflower. It is made from lichen and comes as a solution or as a strip of paper called litmus paper.
What does word indicator mean in chemistry?
Chemical indicator.
How is an indicator defined in chemistry quizlet
A chemical indicator is a substance that undergoes remarkable observable changes when the conditions for its dissolution change. This can be color change, precipitation, bubble formation, temperature change or any other measurable quality.
How is an indicator defined in chemistry examples
A chemical indicator, any substance that gives a visible indication, usually by a color change, of the presence or absence of a threshold concentration of a chemical, such as an acid or an alkali, in a solution. An example is a substance called methyl yellow, which gives an alkaline solution a yellow color. If the acid is added slowly, the solution will remain yellow until all the alkali is neutralized, at which point the color suddenly turns red.
What are indicators used for chemistry studies
Indicators are substances that change color when added to acidic or alkaline solutions. Sunflower, phenolphthalein and methyl orange are commonly used indicators in the laboratory. The sunflower indicator solution turns red in acidic solutions, blue in alkaline solutions, and violet in neutral solutions.
![:brown_circle: :brown_circle:]() What are some examples of the indicators in chemistry?
What are some examples of the indicators in chemistry?
- The pH indicator changes color in solution in a narrow pH range.
- Fluorescein is a kind of adsorption indicator.
- Other types of fluorescent indicators are designed to bind to selected molecules.
- The indicator can be used to determine the end point of the titration.
- Indicators can indicate the presence or absence of a molecule of interest.
What are indicators used for chemistry notes
General Purpose/Use of Chemical Indicators. Chemical indicators are especially useful in chemistry to determine when a reaction has undergone a chemical change or is complete. The idea behind using a chemical indicator is to indicate chemical concentrations by visible signs.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What are the different types of chemical indicators?
What are the different types of chemical indicators?
In other cases, a chemical indicator may be an indicator based on an irreversible reaction. Most chemical indicators are divided into five groups: acid-base, redox, complexometric, adsorption and chemiluminescent indicators.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Why is specificity important in a chemical indicator?
Why is specificity important in a chemical indicator?
Specificity is important for synthetic host molecules to recognize and bind to host molecules. In other cases, a chemical indicator may be an indicator based on an irreversible reaction.
How is turmeric used as a chemical indicator?
The turmeric they use in their lifestyle for cooking can also be used as a chemical indicator. When applied to an acidic environment, it will turn yellow. And as a basic solution, it turns brown.
Which is an example of a pH indicator?
The hydrogen performance indicator (pH) changes color over a narrow range of pH values in a given solution. There are many different pH indicators that display different colors and operate within certain ranges. A classic example is litmus paper.
What is an indicator chemistry
A chemical indicator, any substance that gives a visible indication, usually by a color change, of the presence or absence of a threshold concentration of a chemical, such as an acid or an alkali, in a solution.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What type of compound is indicator?
What type of compound is indicator?
The indicator is a strong acid or base. Indicators are a strong base and a strong acid, because they allow you to tell by color whether a compound is acidic or basic.
![:brown_circle: :brown_circle:]() What is the definition of indicator?
What is the definition of indicator?
Definition of indicator 1: one that indicates: for example 3: an organism or ecological community so closely related to certain environmental conditions that its presence indicates the existence of those conditions.
![:brown_circle: :brown_circle:]() What is the use of indicator?
What is the use of indicator?
Indicator (name) - A device to display the operating status of the system. Indicator (name) (chemical composition) A substance that changes color to indicate the presence of an ion or substance can be used to indicate the completion of a chemical reaction or (in medicine) to indicate a specific test response.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What is the definition of indicator in biology?
What is the definition of indicator in biology?
Definition: A biological indicator refers to organisms, species or communities whose characteristics indicate the presence of certain environmental conditions. Other terms used are indicator organism, indicator plant and indicator species.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What does an indicator do in science?
What does an indicator do in science?
An indicator is a chemical added to a mixture that can detect the presence of a specific chemical. The indicators can show their results by changing color, emitting scents or creating light.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What color is a pH scale?
What color is a pH scale?
The pH scale runs from 14 to 14, each number has its own color. At the bottom of the scale is red, which represents the strongest acidity, and dark blue, on the other side, for 14 and alkalinity. In the middle zone, the pH scale becomes neutral.
![:brown_circle: :brown_circle:]() What is an example of pH?
What is an example of pH?
The pH value is defined as the acidity or alkalinity of a solution. The pH in the example is 7 for distilled water, indicating neutrality.
What is the pH scale?
The pH scale measures the acidity or alkalinity of a substance. The pH scale runs from 14 to 14. The pH value of 7 is neutral. PH below 7 is acidic. PH above 7 is basic. The pH scale is logarithmic, so any total pH value below 7 is ten times more acidic than the next highest value.
What is a pH indicator and what is it used for?
Use of pH indicators. PH indicators are used to determine the approximate pH value of a chemical solution. The PH meter is used for accurate measurements. Alternatively, absorption spectroscopy with a pH indicator can be used to calculate pH according to Beers' law.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() Which pH indicator is the most accurate?
Which pH indicator is the most accurate?
The most accurate indicator is a digital pH meter. This indicator can detect the pH of a substance and display the exact pH value on digital displays. It has a ■■■■■■■■/probe that is dipped into the solution and then the pH is displayed on the screen.
What is pH indicator and how they work?
A pH indicator is a chemical compound that can measure the amount of hydrogen or hydroxide ions in a solution. PH indicators often work by changing color depending on the acidity of the solution.
How can a pH indicator be useful?
PH indicators can be used in a variety of ways, including measuring the pH of farmlands, shampoos, juices and ponds. In addition, pH indicators occur naturally so their presence in plants and flowers can indicate the pH of the soil in which they grow.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Endpoint chemistry
Endpoint chemistry
Difference Between Endpoint and Equivalence Point Titration is used in analytical chemistry to determine acids, bases, reducing agents, oxidizing agents and other substances. Titration can generally occur in reactions such as redox reactions and acid-base reactions. Two major steps are reached in the process known as the end point and the equivalence point.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What is the definition of endpoint in chemistry?
What is the definition of endpoint in chemistry?
In chemistry, the end point is the completion of a chemical reaction. Particularly in titration, this is the point where the titrant should no longer be added, which is usually indicated by a change in indicator color or other measurable change.
What is end point in chemistry?
Or the endpoint (ĕnd′point ′) element 1. One of the two points that indicate the end of the line segment. 2. Chemical composition. The titration point where no titrant should be added. This is determined, for example, by a change in the color of an indicator or by the appearance of precipitation.
Is endpoint the same as equivalence point?
Although the endpoint is generally considered an equivalence point, they are not the same. But since there is only a small difference between the equivalent point and the end point, the same can be considered for the lab. The end point always comes after the equivalence point.
![:brown_circle: :brown_circle:]() What is the definition of endpoint titration?
What is the definition of endpoint titration?
What is the end point. The end point of the titration is the point where the color change occurs. Acids and bases are usually colorless solutions. Therefore, to determine the end of the acid neutralization reaction with a base, an indicator is used that can change the color of the reaction mixture when the pH changes.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() Universal indicator chemistry
Universal indicator chemistry
Experiments with the universal indicator Yamadas are also described in the Journal of Chemical Education. The universal indicator generally consists of water, propanol, phenolphthalein, sodium hydroxide, methyl red, bromothymol blue, sodium bisulfite and thymol blue.
![:brown_circle: :brown_circle:]() How does universal indicator work?
How does universal indicator work?
Therefore, a universal indicator is a mixture of indicators that show a color change in a solution, which interprets how acidic or basic a solution is. The universal indicator can be on paper or in the form of a solution.



