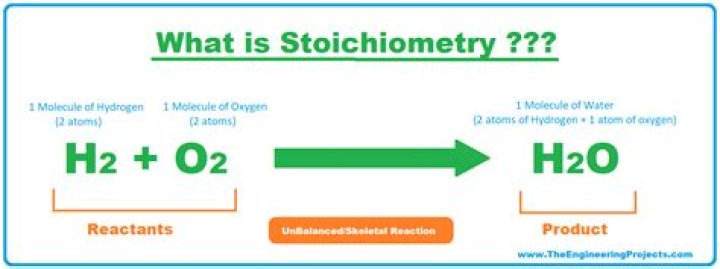
How is stoichiometry used in real life

Sarah Scott
Published Apr 11, 2026
Stoichiometry is at the heart of the production of many things you use in your daily life. Soap, tires, fertilizer, gasoline, deodorant, and chocolate bars are just a few commodities you use that are chemically engineered, or produced through chemical reactions.
When stoichiometry can be used in real situations?
Stoichiometry continues to be useful in many walks of life—a farmer determining how much fertilizer to use, figuring out how fast you have to go to get somewhere in a specific period of time, or just to make conversions between systems like Celsius and Fahrenheit.
How is stoichiometry used in the industry?
Stoichiometry is used in industry quite often to determine the amount of materials required to produce the desired amount of products in a given useful equation.
What can stoichiometry be used for?
Stoichiometry is the field of chemistry that is concerned with the relative quantities of reactants and products in chemical reactions. … In addition, stoichiometry can be used to find quantities such as the amount of products that can be produced with a given amount of reactants and percent yield.What is an example of stoichiometry?
Example – Using Stoichiometric Ratio (Moles) By looking at the coefficients, you can see that for every 1 mole of C6H12O6, 2 moles of CO2 are produced. Using this ratio, you can figure out how many moles of carbon dioxide are made from 2.5 moles of glucose.
How is stoichiometry used in healthcare?
Nurses routinely use stoichiometry to convert the drip ratios of medication to the prescribed dosages. They use a persons weight and conversion factors to determine the correct dosage of medicine to give. If the stoichiometry calculations are done incorrectly the patient can potentially be harmed.
How can stoichiometry be used in the future?
Stoichiometry predicts the amount of product produced For example, we used stoichiometry to determine how many s’more “reactants” we would need to make 10 s’mores. We can also use stoichiometry to predict how much product we’ll get with the amount of each reactant we have.
What is stoichiometry used for Apex?
stoichiometry is important for determining the ratio of products to reactants in a chemical reaction. Therefore the use of this would allow the chemists to accurately predict an expected mass of the product with given quantities.What are some of the real life applications of the mole?
In chemistry, the mole is a unit used to talk about atoms. It is similar to other units we use everyday. For example, you might walk into the local doughnut shop and order a dozen doughnuts. In doing so, you know that you will get 12 of these snacks and the clerk knows to give you 12.
Why do scientists use stoichiometry?To manipulate chemical reactions on a large scale, scientists use stoichiometry to quantify those reactions and make sure that there are just the right amount of reactants and products. … Stoichiometry gives us the quantitative tools to figure out the relative amounts of reactants and products in chemical reactions.
Article first time published onDo pharmacists use stoichiometry?
As shown in my first sample from the workplace, stoichiometry problems are a typical part of a pharmacist’s daily routine. … Pharmacists use the mole and various calculations that use this value to mix chemicals that form powders, tablets, and ointments.
How is mole used?
The mole can be used to determine the simplest formula of a compound and to calculate the quantities involved in chemical reactions. When dealing with reactions that take place in solutions, the related concept of molarity is useful. Molarity (M) is defined as the number of moles of a solute in a litre of solution.
How is the mole used by chemists?
A mole is a very important unit of measurement that chemists use. A mole of something means you have 602,214,076,000,000,000,000,000 of that thing, like how having a dozen eggs means you have twelve eggs. Chemists have to measure using moles for very small things like atoms, molecules, or other particles.
How do we use moles in medicine?
The mole (particularly millimoles and molarity) is a commonly used unit in medicine. For example, concentrations of intravenous solutions and drugs are expressed in terms of moles.
Why does an airbag contain KNO3 and SiO2?
The purpose of the KNO3 and SiO2 is to remove the sodium metal (which is highly reactive and potentially explosive) by converting it to a harmless material.
Which gas is used in car airbags?
Either nitrogen or argon gas is used to inflate an airbag. Both of these gases are non-toxic. Immediately after a deployment, “smoke-like” residue will be present in the air. Most of this residue is talcum powder that is used as a lubricant to help the airbag deploy smoothly.
Why is a decomposition reaction used for airbags?
CRASHES trip sensors in cars that send an electric signal to an ignitor. The heat generated causes sodium azide to decompose into sodium metal and nitrogen gas, which inflates the car’s air bags. Under normal circumstances, this molecule is quite stable. If heated, though, it will fall apart.
Why is stoichiometry solution important?
Stoichiometry is important because it shows the relative amount of each reactant needed for a reaction to proceed, and helps determine the expected amount of products. Stoichiometry is important in analytical chemistry, analysis methods, and understanding chemical reactions.
What kind of reaction is symbolized by AB a B?
A decomposition reaction is often symbolized as AB → A + B; where AB is the substrate, and A and B are the products. Different types of decomposition reactions may also be referred to as digestion, hydrolysis, breakdown, and degradation reactions.
What is the importance of stoichiometry in agriculture?
Altered stoichiometry in agricultural systems (example 1) can affect not only crop yield and quality but also the interactions between plants and their pollinators, pests and pathogens. Human activities have also altered stoichiometry in coastal ecosystems (example 2).
What is stoichiometry in your own words?
Stoichiometry is exactly that. It is the quantitative relation between the number of moles (and therefore mass) of various products and reactants in a chemical reaction. Chemical reactions must be balanced, or in other words, must have the same number of various atoms in the products as in the reactants.
How is stoichiometry used to calculate the mass of a substance?
By definition, carbon-12 has a molar mass of 12 g/mol. Thus, to calculate the stoichiometry by mass, the number of molecules required for each reactant is expressed in moles and multiplied by the molar mass of each to give the mass of each reactant per mole of reaction.
Why is the moles used to measure the atom?
Why We Use Moles The answer is that moles give us a consistent method to convert between atoms/molecules and grams. It’s simply a convenient unit to use when performing calculations.
Why do we use grams instead of moles?
You need to convert using the weight. Grams depends on the molecule itself, oxygen weighs more than hydrogen. But using moles gets rid of the mass concern, because a mole represents a specific amount of molecules, independant of weight.
Are moles blind?
Moles are often thought to be blind when in fact they can see; they are, though, colorblind and have poor vision only adapted to recognize light. To find food and to navigate the dark underground, moles rely on their keen sense of smell and touch. … Moles are small, burrowing mammals who live underground.
Why do we celebrate Mole Day?
Celebrated annually on October 23 from 6:02 a.m. to 6:02 p.m., Mole Day commemorates Avogadro’s Number (6.02 x 10²³), which is a basic measuring unit in chemistry. Mole Day was created as a way to foster interest in chemistry.
What is the importance of the mole concept in chemistry select all that apply?
What is the importance of the mole concept in chemistry? Select all that apply. It allows chemists to study and measure substances within the subatomic world. It allows chemists to change grams to the number of particles through the mole, even though particles cannot be seen.