How is carbon monoxide formed in a blast furnace

Emily Cortez
Published Apr 19, 2026
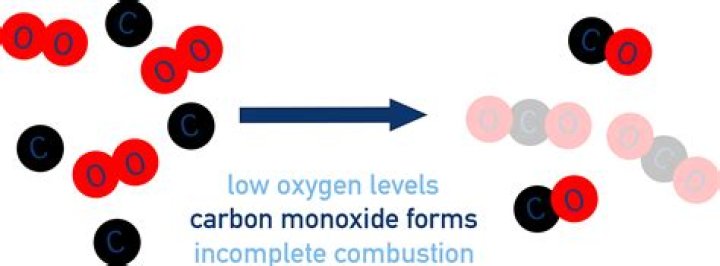
In the blast furnace, there are several chemical reactions taking place; that eventually result in the desired product (iron) being extracted. The coke (carbon) burns with oxygen to produce carbon dioxide. This reaction is exothermic. The CO2 then reacts with more coke to give carbon monoxide.
What is the reaction that happens in the blast furnace?
The reactions of the blast furnace involve 1) combustion of the fuel and its conversion into carbon monoxide, 2) reduction of the ore, and 3) formation of slag. A reaction such as FeO + CO = Fe + CO2 can occur in both the forward and backward direction under conditions existing somewhere in the blast furnace.
How is slag formed in a blast furnace?
Blast furnace slag (BFS) is a by-product from iron production in blast furnaces, which are fed by a mixture of iron-ore, coke and limestone. In the process, the iron ore is reduced to iron while all remaining materials form the slag, which is tapped off as a molten liquid and cooled.
What gas is produced in a blast furnace?
Steel production gas typeCoke GasBlast Furnace GasInputCoalCoke & iron oreOutputCokePig ironHydrogen %50-70%5%Methane %25-30%–Why is coke added to the blast furnace?
Publisher Summary. Metallurgical coke, along with iron ore and limestone, is layered into a blast furnace to convert the iron ore to metallic iron. Coke, which is mostly carbon, reacts with the blast air to produce carbon monoxide, which, in turn, reacts with the iron oxide to produce carbon dioxide and metallic iron.
How is pig iron produced in the blast furnace?
Blast furnaces produce pig iron from iron ore by the reducing action of carbon (supplied as coke) at a high temperature in the presence of a fluxing agent such as limestone.
How much co2 does a blast furnace produce?
But blast furnaces produce an incredible amount of carbon dioxide. A recent study showed that 252.5 million tons of CO2 was produced in the period from 2005-2008. Putting it another way, blast furnaces produce between four and seven percent of all the man-made CO2 in the world.
Why is limestone added to the blast furnace?
Limestone is also used to remove impurities from the blast furnace when making iron. The impurities are mostly silicon dioxide (also known as sand). The calcium carbonate in the limestone reacts with the silicon dioxide to form calcium silicate (also known as slag).Why Nitrogen gas is used in blast furnace?
Nitrogen gas is used for blanketing purpose. Nitrogen blanketing is used to protect flammable or explosive solids and liquids from contact with air.
What is the composition of slag?The primary components of iron and steel slag are limestone (CaO) and silica (SiO2). Other components of blast furnace slag include alumina (Alsub>2O3) and magnesium oxide (MgO), as well as a small amount of sulfur (S), while steelmaking slag contains iron oxide (FeO) and magnesium oxide (MgO).
Article first time published onWhy coal is not used in blast furnace?
A blast furnace is a reactor which produces an impure form of molten iron, termed ‘hot metal’, from ferrous bearing ores. 1. Metallurgical coke, ferrous bearing feeds and fluxes charged via the furnace top. … Coal cannot be directly charged via the furnace top, it must first be transformed to coke.
Why do we use coke not coal for making iron?
Coke is used as a fuel and a reducing agent in melting iron ore. It is produced by baking coal until it becomes carbon by burning off impurities without burning up the coal itself. When coke is consumed it generates intense heat but little smoke, making it ideal for smelting iron and steel.
Why coke is not used as a fuel?
Answer: because fuel has different properties than coke as fuel can help a car move and coke can’t do do that as it does not have the chemicals to complete that task …
What happens to carbon dioxide in the blast furnace?
The blast furnace Reactions in which oxygen is removed are called reduction reactions. In this reaction, the iron(III) oxide is reduced to iron, and the carbon is oxidised to carbon dioxide.
Does smelting release carbon monoxide?
In copper smelting process, first, the carbon (C) combusts with oxygen (O2) in the air to produce carbon monoxide (CO). Second, the carbon monoxide reacts with the ore and removes one of its oxygen atoms, releasing carbon dioxide. Hence, it does not release lethal quantity of CO in the environment.
What materials are used in a blast furnace to produce pig iron?
The three main raw materials used to make pig iron (which is the raw material needed to make steel) for primary steel production in a blast furnace are the processed iron ore, coke (residue left after heating coal in the absence of air, generally containing up to 90% carbon) and limestone (CaCO3) or burnt lime (CaO), …
Why is pig iron called pig iron?
The iron produced is called “pig iron” because originally the molten iron was poured into sand pits and the resulting solid iron resembled a pig! Another story is that the system looked like piglets feeding from a sow.
What is the end product of blast furnace?
A blast furnace is a type of metallurgical furnace used for smelting to produce industrial metals, generally pig iron, but also others such as lead or copper. … The end products are usually molten metal and slag phases tapped from the bottom, and waste gases (flue gas) exiting from the top of the furnace.
What is the role of caco3 in the blast furnace?
Limestone is calcium carbonate (CaCO3) and it is added to the blast furnace to remove the impurities in the iron ore, as mentioned above. Calcium carbonate is decomposed by heat in the furnace to give calcium oxide (quicklime) and carbon dioxide.
Why blast furnace is called blast furnace?
Special nozzles called “tuyeres” are used to put the air in the furnace. The nozzles are at the bottom of the furnace. This process is called “blasting”. It is why it is called a “blast furnace”.
How is limestone removed from the molten steel in a blast furnace?
The limestone is added to convert this into slag which melts and runs to the bottom. The heat of the furnace decomposes the limestone to give calcium oxide. This is an endothermic reaction, absorbing heat from the furnace.
What is blast furnace slag in concrete?
Slag cement is a hydraulic cement formed when granulated blast furnace slag (GGBFS) is ground to suitable fineness and is used to replace a portion of portland cement. It is a recovered industrial by-product of an iron blast furnace.
Which of the following is separated as slag during extraction Fe in blast furnace?
Carbon monoxide act as reducing agent during the reaction in blast furnace and lime stone decomposes in the hot furnace forming calcium oxide. The molten iron is then tapped from the bottom of the furnace. Further, calcium oxide react with SiO2 to form a slag.
What mineral that produced slag?
Iron and steel slag, also known as ferrous slag, is produced by adding limestone (or dolomite), lime and silica sand to blast furnaces and steel furnaces to strip impurities from iron ore, scrap and other ferrous feed materials and to lower the heat requirements of the iron- and steelmaking processes.
How is coke obtained?
Coke is obtained by destructive distillation of charcoal or coal in the furnace Destructive distillation is the chemical process involving the decomposition of coal or charcoal by heating to a high temperature in the absence of air or in the presence of limited amounts of oxygen.
Which oxide is reduced in blast furnace?
In a blast furnace, iron oxide is reduced by Carbon Monoxide. A substance called coke (carbon) is added along with oxygen(O2) to form CO. The Blast furnace is used to produce pig iron from iron ore.
Which oxygen is used for blast furnace operation?
More than half the world’s steel is produced in the basic oxygen process (BOP), which uses pure oxygen to convert a charge of liquid blast-furnace iron and scrap into steel.
What is difference between coal and coke?
Coal is a lustrous, black fossil fuel that includes impurities, generates smoke, and generates less heat than coke when burned. Coke is a dingy, black coal waste that burns hotter and cleaner. … Coke is a fuel made from mineral coal that has been calcined or dry distilled.
Is coke same as coal?
What is the difference between coal and coke fuel? Coal is a shiny, black fossil fuel that contains impurities, emits smoke when burned, and produces less heat than coke. Coke is a dull, black byproduct of coal that burns hotter and cleaner. … By the 1880s, most plants used coal’s byproduct, coke, to produce steel.
Can steel be made without coal?
Now, nearly all new steel globally is produced using iron oxide and coking coal. Coking coal is usually bituminous-rank coal with special qualities that are needed in the blast furnace. While an increasing amount of steel is being recycled, there is currently no technology to make steel at scale without using coal.
Does Diet coke still exist?
The original flavor was still available for a limited time via Amazon.com during the time of the new flavor rollout, but has since been discontinued, along with Diet Coke Lime. It still currently remains available from Coca-Cola Freestyle machines.