How does molar mass affect osmotic pressure

Mia Russell
Published Apr 12, 2026
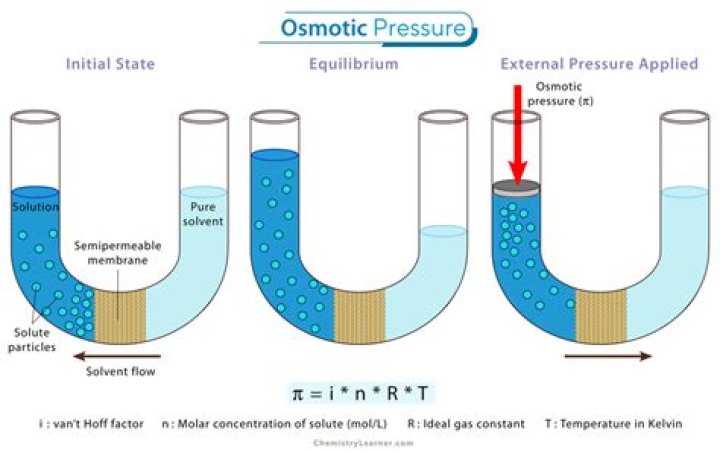
The osmotic pressure of a solution is proportional to the molar concentration of the solute particles in solution.
What is the relation between osmotic pressure and molecular weight?
The flow of solvent that occurs due to a concentration gradient across the membrane is called osmosis. According to Equation 5, the molecular weight of a solute can be obtained by plotting osmotic pressure divided by c versus concentration and extrapolating the data back to c=0.
How does molar concentration affect osmosis?
Concentration gradient – The movement of osmosis is affected by the concentration gradient; the lower the concentration of the solute within a solvent, the faster osmosis will occur in that solvent. Light and dark – They are also factors of osmosis; since the brighter the light, the faster osmosis takes place.
What factors affect osmotic pressure?
Osmotic pressure is affected by concentration and temperature. Concentration of solute and temperature each affect the amount of pressure created by the movement of water across a membrane. Higher concentrations and higher temperatures increase osmotic pressure.Why osmotic pressure is good for determining the molar mass?
(a) Measurement of osmotic pressure method is preferred for the determination of molar masses of macromolecules such as proteins and polymers because the magnitude of osmotic pressure is large even for very dilute solution and it can be measured at room temperature.
How the molecular mass is determined by osmotic pressure method in polymer?
The osmotic pressure is the minimum pressure that has to be applied to a polymer solution to prevent a solvent to flow through a semipermeable membrane that separates pure solvent from polymer dissolved in the same solvent. Osmometry is frequently used to determine the number-average molecular weight of polymers.
Which type of molar mass can be determined using osmotic pressure method?
The molar mass of high molar mass solute can be calculated by osmotic pressure.
What controls the osmotic pressure?
Since osmotic pressure is regulated by the movement of water across membranes, the volume of the fluid compartments can also change temporarily. Because blood plasma is one of the fluid components, osmotic pressures have a direct bearing on blood pressure.What causes osmotic pressure to increase or decrease?
loss of electrolytes (salt), the osmotic pressure of the extracellular fluids becomes higher than in the cells. Since water passes from a region of lower to a region of higher osmotic pressure, water flows out of the cells into the extracellular fluid, tending to lower its osmotic pressure and increase…
What happens if osmotic pressure is high?(c) When cells are placed in a concentrated salt solution with an osmotic pressure greater than that of the intracellular fluid, the rate of flow of water out of the cells is greater than the rate of flow into the cells. The cells shrivel and become so deformed that they cannot function.
Article first time published onHow does mass affect osmosis?
During osmosis substances move from an area of high pressure to an area of low pressure. Therefore, when a chip is placed in a concentrated sucrose solution, it will lose mass because the chip has a higher concentration of water than the sucrose solution.
How the solute concentration affects osmotic pressure?
The osmotic pressure driving water across an impermeable barrier increases with the difference in solute concentrations on either side of the barrier. … Osmotic pressure depends only on the number of solute particles, not on their composition.
What are the advantages of osmotic pressure?
Advantage of using osmotic pressure as compared to other collegative properties: (i) The equilibrium is established very quickly. Hence, results are obtained in a very short time. (ii) The concentration of the solution does not change during determination of osmotic pressure.
What are the two reasons that the molecular masses of polymers are determined by osmotic pressure method and not by measuring other colligative properties?
The molecular masses of polymers are determined by osmotic pressure method and nor by measuring other colligative properties because of the following two reasons: (i) Osmotic pressure is measured at room temperature so the molecule does not dissociate. (ii) Osmotic pressure has appreciable value.
Why measurement of osmotic pressure method is preferred for the Determination of molar mass of macromolecules such as protein and polymers?
Measurement of osmotic pressure method is preferred for the determination of molar masses of macromolecules such as proteins and polymers because this method has the advantage that is use molarities instead of molalities . Secondly , the changes observed in these properties are very small .
How can molar mass of a substance can be determined from the measurement of osmotic pressure of a solution?
Osmotic pressure (π) is defined as the extra pressure that must be applied to the solution side in order to prevent the flow of solvent molecules into it through a semipermeable membrane. Thus, knowing WB , T, π and V molecular mass of the solute, MB can be calculated.
Why osmotic pressure is a better criteria for measuring the molecular mass of macromolecules as compared to elevation of boiling point or depression of freezing point?
As compared to the colligative properties like elevation in boiling pint and depression in freezing point, osmotic pressure is a good criterion for finding the molecular mass of macromolecules. This is because of the fact that, osmotic pressure is the only colligative property which can be measured at room temperature.
How does osmotic pressure affect cells?
Osmotic pressure is of vital importance in biology as the cell’s membrane is selective toward many of the solutes found in living organisms. When a cell is placed in a hypertonic solution, water actually flows out of the cell into the surrounding solution thereby causing the cells to shrink and lose its turgidity.
How does albumin affect osmotic pressure?
serum albumin, protein found in blood plasma that helps maintain the osmotic pressure between the blood vessels and tissues. … Transfusions of serum albumin are used to combat shock and whenever it is necessary to remove excess fluid from the tissues.
How does osmotic pressure depends on temperature and atmospheric pressure?
Osmotic pressure is driven by the tendency of the solute molecules that are not glueing together to take up as much space as they can get. … All else staying equal, this gives a direct proportional relationship between temperature and pressure.
Which of the following would result in no change in osmotic pressure?
Which of the following would result in NO change in osmotic pressure across a membrane? Water is moving with its concentration gradient. The solutes can diffuse through the pores and the concentration of solutes is the same on both sides of the membrane.
Which has maximum osmotic pressure?
So at 1M , (NH4)3PO4 will have the highest value of osmotic pressure .
How is osmotic pressure maintained in the body?
Mammalian systems have evolved to regulate osmotic pressure by managing concentrations of electrolytes found in the three major fluids: blood plasma, extracellular fluid, and intracellular fluid. Water movement due to osmotic pressure across membranes may change the volume of these fluid compartments.
What happens when pressure greater than osmotic pressure is applied on the solution side separated from solvent by a semi permeable membrane?
What will happen if pressure greater than the osmotic pressure is applied on the solution separated by a semi-permeable membrane from the solvent? It will result into reverse osmosis, i.e., there will be net flow of the solvent from the solution to the solvent.
Why osmotic pressure is positive and osmotic potential is negative?
Osmotic pressure is a hydrostatic pressure exerted to the solution to prevent the flow of water through the semi permeable membrane. … As a solution will always have some solute, the amout of water will be lesser than pure water. Thus the osmotic potential of a solution will always be negative (value less than zero).
Does osmosis increase mass?
When the chips were put in distilled water they gain mass because the chips gain water from the surrounding solution due to osmosis. The process of osmosis causes a net flow of water across the semi permeable membrane, from a solution with a high water concentration to one with a lower water concentration.
What factors affect the rate of osmotic movement of water?
The rate of osmosis varies with a number of factors, including temperature, pressure, and the difference in solute concentrations between two solutions separated by a selectively permeable membrane.
What is osmosis which factor affects osmosis in a cell?
Osmosis is movement of only water molecules through a semi permeable membrane from their region of higher concentration to their region of lower concentration. The factors that affect the rate of osmosis are concentration of cell sap, the flow time rate and temperature.
What factors affect osmotic pressure?
Osmotic pressure is affected by concentration and temperature. Concentration of solute and temperature each affect the amount of pressure created by the movement of water across a membrane. Higher concentrations and higher temperatures increase osmotic pressure.
Does osmotic pressure depends on molarity?
The osmotic pressure of a solution is proportional to the molar concentration of the solute particles in solution.
How does molarity affect osmosis?
The concentration of a solute affects the rate of osmosis over time, in a way where, the higher the concentration of a solute, the faster the rate of osmosis. This happens because, in a semi-permeable membrane the water is the only through that can move through. … That results in the rate of osmosis being faster.