How does calorimetry work

David Jones
Published Apr 18, 2026
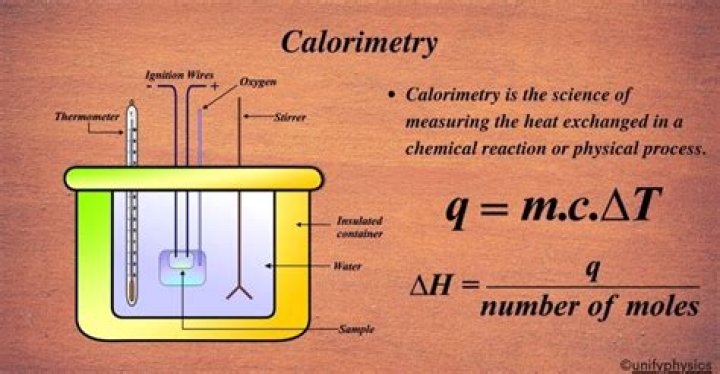
A typical calorimeter works by simply capturing all the energy released (or absorbed) by a reaction in a water bath. … Thus by measuring the change in the temperature of the water we can quantify the heat (enthalpy) of the chemical reaction.
What is the process of calorimetry?
Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. By knowing the change in heat, it can be determined whether or not a reaction is exothermic (releases heat) or endothermic (absorbs heat).
What is C in calorimetry?
The heat capacity, C, of a substance is the amount of heat required to raise the temperature of a given quantity of the substance by 1 degree. … The heat capacity of the calorimeter is the quantity of heat absorbed by the calorimeter for each 1°C rise in temperature.
How does a calorimetry experiment work?
Simple calorimeters are made with a metal container of water, positioned above a combustion chamber. A thermometer is used to measure the heat change in the amount of water. … The calorimeter measures the mass of the liquid along with the temperature change, to determine the amount of energy change.How do you tell if heat is absorbed or released?
Enthalpy of a reaction is defined as the heat energy change ( Δ H ΔH ΔH ) that takes place when reactants go to products. If heat is absorbed during the reaction, Δ H ΔH ΔH is positive; if heat is released, then Δ H ΔH ΔH is negative.
What is calorimetry in biochemistry?
Calorimetry is a general term referring to the measurement of the quantities of heat evolved or absorbed in physical or chemical processes. Since most chemical processes do occur with measurable heat effects, calorimetry can be used to study a wide variety of chemical systems.
How does calorimeter measure energy?
The calorimeter traps all the heat from a chemical reaction, we measure the effect of that heat on the temperature of water in the calorimeter, and we can then calculate the heat energy released by the reaction. The calorimeter is an insulated container, in which we place a measured mass of water.
What does Q mean in calorimetry?
Heat capacity (C) is the amount of heat (q) required to raise the temperature of an object one degree Celsius. The units for heat capacity are J/oC (the unit is read as Joules per degree Celsius).How do you do a calorimetry lab?
Place the thermometer in the calorimeter cup and record the temperature for 3 readings at 30 second intervals. Lift the lid of the calorimeter and drop the pieces of magnesium in, mixing continuously. Record the temperature every 30 seconds until 10 minutes have elapsed.
Does a calorimeter release heat?The thermal energy change accompanying a chemical reaction is responsible for the change in temperature that takes place in a calorimeter. If the reaction releases heat (qrxn < 0), then heat is absorbed by the calorimeter (qcalorimeter > 0) and its temperature increases.
Article first time published onWhat does a positive Q mean?
When heat is absorbed by the solution, q for the solution has a positive value. This means that the reaction produces heat for the solution to absorb and q for the reaction is negative. … This means that the reaction absorbs heat fron the solution, the reaction is endothermic, and q for the reaction is positive.
What does Q MCAT stand for?
Q = Heat energy (in Joules, J) m = Mass of a substance (kg) c = Specific heat (J/kg∙K) ∆T = Change in temperature (Kelvins, K)
Is heat released when water freezes?
Changes of state involve a solid melting, a liquid freezing, a liquid boiling or a gas condensing. When steam, which is gaseous water, condenses, heat is released. Likewise when liquid water freezes, heat is given off. In fact heat must be continually removed from the freezing water or the freezing process will stop.
Where does absorbed energy go?
Q: If energy can’t be destroyed during a chemical reaction, what happens to the energy that is absorbed in an endothermic reaction? A: The energy is stored in the bonds of the products as chemical energy. In an endothermic reaction, the products have more stored chemical energy than the reactants.
Why bomb calorimeter is called bomb?
Gases at high pressures are frequently used, hence the name. In 1878, Paul Vieille (1854–1934) developed the first bomb calorimeter which was used for measuring heats of explosion at the French service of explosives in Paris. However, this bomb was attributed by many authors to M. Berthelot (1827–1907).
How is calorimetry used in industry?
Calorimeters are useful in various industries and academic settings, an industrial pilot plant can use a DSC to determine a change in a products formula and how it affects the formula itself. Oxygen bomb calorimeters are useful in food testing laboratories to determine the amount of heat (calories) in food.
Who discovered calorimetry?
Lavoisier and Laplace are generally credited with being the inventors of direct calorimetry and the first to use this method for the measurement of animal heat (32). Their calorimeter was called an ice calorimeter because the heat evolved was measured by the amount of ice that it caused to melt.
Why do you put a lid on the Styrofoam cup?
Coffee cups, especially those made of Styrofoam, are effective calorimeters because they hold in the heat. … The lid should be flat and make a good seal when placed inside the lip of the cup. Cardboard acts as a better insulator than plastic lids do.
How do you calculate calorimetry?
- Q = heat evolved (equal to heat absorbed − heat released) in joules (J)
- m = mass in kilograms (kg)
- c = specific heat capacity in J/kg⋅°C (or J/kg⋅K)
- ∆T = temperature change in °C (or K)
What is calorimetry a level chemistry?
Calorimetry is the tool for computation of exchanged heat energy among chemical reactions. The measurement of heat flow using temperature change are performed using bomb calorimetry. Measurement of physical changes are performed using ice calorimetry.
What does W mean in Chem?
Weight concentration of a solution is expressed as % w/w. Like before, this stands for weight per weight. In this case, the volume of each chemical is disregarded and only the weight is used.
What is W thermochemistry?
Work Energy used to cause an object that has mass to move. w = F × d • Energy is the ability to do work or transfer heat. … Thermochemistry is the study of chemical reactions and the energy changes that involve heat.
What does W stand for in Thermo?
Section Summary. The first law of thermodynamics is given as ΔU = Q − W, where ΔU is the change in internal energy of a system, Q is the net heat transfer (the sum of all heat transfer into and out of the system), and W is the net work done (the sum of all work done on or by the system).
Why is calorimetry not accurate?
The biggest source of error in calorimetry is usually unwanted heat loss to the surroundings. This can be reduced by insulating the sides of the calorimeter and adding a lid.
What affects calorimetry?
Calorimetry measures enthalpy changes during chemical processes, where the magnitude of the temperature change depends on the amount of heat released or absorbed and on the heat capacity of the system.
Why is calorimetry important?
Calorimetry is used to determine the heat transfer between two states or environments caused by chemical and physical changes. Calorimetry is important because it is used to figure out changes in temperature based on how much heat is gained or given by a system in a reaction.
What does ∆ s mean?
∆S is the change in entropy (disorder) from reactants to products. R is the gas constant (always positive) T is the absolute temperature (Kelvin, always positive) What it means: If ∆H is negative, this means that the reaction gives off heat from reactants to products.
Is Delta H the same as Q?
Q is the energy transfer due to thermal reactions such as heating water, cooking, etc. anywhere where there is a heat transfer. You can say that Q (Heat) is energy in transit. Enthalpy (Delta H), on the other hand, is the state of the system, the total heat content.
Can you have a negative delta T?
The delta-T can be positive (Temp increases) or negative (Temp decreases).
What does the Q mean in Q mcAT?
Re: q equation q=mcAT is for when you are given the mass and q=ncAT is used when you are given moles.
What is the C in Q MC T?
Q=mcΔT Q = mc Δ T , where Q is the symbol for heat transfer, m is the mass of the substance, and ΔT is the change in temperature. The symbol c stands for specific heat and depends on the material and phase. The specific heat is the amount of heat necessary to change the temperature of 1.00 kg of mass by 1.00ºC.