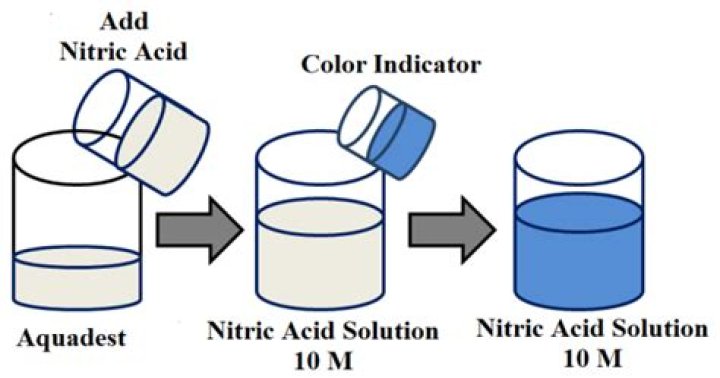
How do you make 1 solution of nitric acid

Sarah Scott
Published Apr 09, 2026
Dissolving 1.5 grams of nitric acid in 150 grams of water gives 1%. This is the same as dissolving 1 mL of nitric acid in 150 mL of water. The extra 50 mL of the solution is to be poured in another container. It is also 1% of nitric acid.
How do you make a 5% nitric acid solution?
The volume of 7.14 g of concentrated HNO3 can be obtained from the density V = 7.14 g / 1.42 = 5.03 mL. If you then measure 5.03 mL of the concentrated HNO3 and dilute to 100 mL with water, you would have the required 5% solution.
What must be dissolved in water to make nitric acid?
Nitric acid is made by reaction of nitrogen dioxide (NO2) with water. The net reaction is: 3 NO2 + H2O → 2 HNO3 + NO. Normally, the nitric oxide produced by the reaction is reoxidized by the oxygen in air to produce additional nitrogen dioxide.
Can you dilute nitric acid with water?
Add the concentrated acid slowly to the water with stirring. Dissolution of the concentrated acid will generate heat. 5. With distilled water from the wash bottle, rinse the remaining acid from the small measuring cylinder into the solution in the large beaker.How do you make 0.1 N nitric acid?
Equivalent weight of nitric acid : Volume prepared from 6.3 g of nitric acid: 1 Litre of 0.1N nitric acid solution will be prepared from 6.3g of nitric acid.
How do you dilute a solution?
Dilution is the process of decreasing the concentration of a solute in a solution, usually simply by mixing with more solvent like adding more water to the solution. To dilute a solution means to add more solvent without the addition of more solute.
How do you make a 10% nitric acid solution?
3.2 Pour about 75.0 ml of distilled water into a 250 ml capacity beaker. 3.3 Transfer slowly, through sidewalls of the beaker, 10.6 ml of Nitric Acid LR into above said beaker, while gentle mixing, by using a glass rod. 3.4 Transfer prepared solution into a 100 ml volumetric flask by using a glass funnel.
How do you distill nitric acid?
The simplist and most used preparation of nitric acid in the lab is by adding concentrated sulfuric acid to a dry nitrate salt, usually, potassium or sodium nitrate. This mixture is heated to melt the pasty mixture and distill the nitric acid.Which one is diluted?
Concentrated solutionDilute solutionA liquid with a high solute concentration is called a concentrated solution.A liquid with a less solute concentration is called a dilute solution.
How do you make nitric acid from household items?Dissolve 80 grams (2.8 oz) of nitrate salt in 50 millilitres (1.7 fl oz) of water. Start by portioning out your nitrate salt into a small glass mixing container. Then, pour in all of the water at once. Swirl the mixture around inside the container to help it dissolve faster.
Article first time published onHow is nitric acid made?
Nitric acid is made by the reaction of nitrogen dioxide (NO2) with water. Normally, the nitric oxide produced by the reaction is re-oxidized by the oxygen in air to produce additional nitrogen dioxide. Adding sulfuric acid to a nitrate salt and heating the mixture with an oil bath can make almost pure nitric acid.
How do you make a 1M HCl solution?
- 1M HCl: add 1mol/12M = 83 ml conc. HCl to 1L of water or 8.3ml to 100ml.
- 2M HCl: add 2mol/12M = 167 ml conc. HCl to 1L of water or 16.7ml to 100ml.
How do you make 1M Sulphuric acid?
- The Molecular weight of is 98 g/mol.
- Next we add 98g of conc. …
- As Acid is a liquid,density should be taken into account.
- Density =Mass/volume.
- Density given on the bottle of Sulphuric acid is 1.84 g/ml.
How do you make 6M nitric acid?
For example, to make 500 mL of 6M HCl, use 250 mL of concentrated acid and slowly dilute to 500 mL with water.
How do you calculate the percentage of nitric acid?
Find the total mass of the nitric acid HNO3 Then divide the mass of each atom by the total mass to find the percentage.
What is 10% nitric acid?
A 10% nitric acid solution is commonly used as a cleaning product while sanitizing food and beverage processing equipment and also for metal surface treatment. A weak solution of nitric acid is also used by woodworkers to “pop” the grain of curly maple.
What is the chemical formula for nitric acid?
A nitrogen oxoacid of formula HNO3 in which the nitrogen atom is bonded to a hydroxy group and by equivalent bonds to the remaining two oxygen atoms. Nitric acid (HNO3), also known as aqua fortis (Latin for “strong water”) and spirit of niter, is a highly corrosive mineral acid.
How do you make a 2 percent nitric acid solution?
For a 2% solution the final weight of solution will be 13.4/. 02=670 grams. Therefore add 670–13.4=656.6 g of H2O, giving 670 g of 2% HNO3.
How do you calculate dilute acids?
Steps to dilute strong acid Use C1V1 = C2V2 relationship. Then separate the required volume from high concentrated solution. Add that acidic solution to water slowly for diluting until required volume of dilute acid is completed.
How do you dilute half?
If you ask someone to dilute a sample in half, pretty much everyone will do it the same way – add an equal volume of sample to an equal volume of diluent, whether that’s 1 mL to 1 mL or 100 µL to 100 µL.
How can a solution be dilute or concentrated?
A solution can be dilute or concentrated. A dilute solution is a solution that has very little solute in the solvent. … A concentrated solution is a solution where the solvent has a lot of solute in the solution.
How do you make a solution?
You prepare a solution by dissolving a known mass of solute (often a solid) into a specific amount of a solvent. One of the most common ways to express the concentration of the solution is M or molarity, which is moles of solute per liter of solution.
How do you make a liquid solution from a stock?
A stock solution is prepared by weighing out an appropriate portion of a pure solid or by measuring out an appropriate volume of a pure liquid, placing it in a suitable flask, and diluting to a known volume. Exactly how one measure’s the reagent depends on the desired concentration unit.
How do you make a standard solution from a stock solution?
Dilute the stock solution in distilled water to give working concentrations of 5, 10, 20, 30, 40, 50 mg/L. To prepare 50 ml of the working standards 5, 10, 20, 30, 40, 50 mg/L respectively, weigh 2.5, 5, 10, 15, 20 and 25 g of stock solution into 50 ml volumetric flasks and make up to the volume with distilled water.
How do I make aqua regia?
Fast Facts: Aqua Regia Aqua regia is a corrosive acid mixture made by combining nitric acid and hydrochloric acid. The usual ratio of acids is 3 parts hydrochloric acid to 1 part nitric acid. When mixing the acids, it is important to add the nitric acid to the hydrochloric acid and not the other way around.
What household item has nitric acid?
AlkalisProductsAcidsProductsSulfuric acid Hydrochloric acid Sodium bisulfate Sodium hypochlorite Acetic Acid Nitric acidDrain cleaner Toilet bowl cleaner House bleach (low concentration) Descalers
How is ammonia converted into nitric acid?
In general, the process for nitric acid manufacture consists of preheating the air, mixing it with ammonia gas, passing it over a platinum catalyst, extracting the heat produced from this reaction by using it to preheat the incoming air and then cooling further, adding secondary air and then absorb- ing the nitrogen …
How is nitric acid prepared in laboratory?
Nitric acid is prepared in the laboratory by heating a nitrate salt with concentrated sulphuric acid. Vapours of nitric acid are condensed to a brown liquid in a receiver cooled under cold water.
How do you make a 1 M solution?
Molar solutions Molarity (M) means the number of moles of solute per liter of solution. To prepare a 1 M solution, slowly add 1 formula weight of compound to a clean 1-L volumetric flask half filled with distilled or deionized water. Allow the compound to dissolve completely, swirling the flask gently if necessary.
How do you make 1 HCL out of 37?
Calculations: Stock bottle of 37% HCL. 37 ml of solute/100 ml of solution. Therefore add 8.3 ml of 37% HCL to 1 liter of D5W or NS to create a 0.1N HCL solution.
How do you dilute a 1M solution to 0.1 M?
Take 1 part of your stock solution and add 9 parts of solvent (usually water but sometimes alcohol or other organic solvent). In all cases you are diluting by the same factor. The concentration of the resulting solution is 1M /10 = 0.1M where 10 is the dilution factor.