Ch3no2 Lewis Structure - How To Discuss

Ava Wright
Published Apr 28, 2026
Ch3no2 Lewis Structure
Lewis Diagram for Nitron, CH3NO2 !!!! Help? 3
Do your best to make the outline clear. Thank you very much
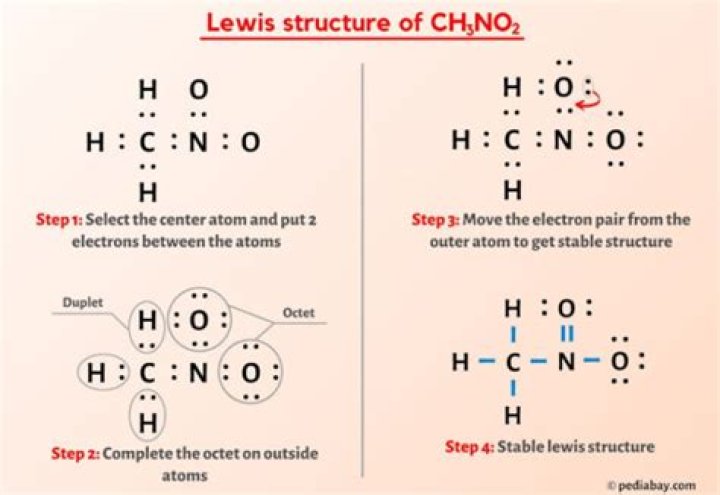
You have to count 24 electrons and C, N and O following the byte principle.
Yes
...... | ..... //
H C N ....... The structure of the second resonance (exchange of N = O and NO).
...... | .....
Yes
The regular charge of single bond O is 1, while the regular charge of N is +1. The rest of the elements have a regular charge, e.g. It is very likely that the resonance structure represents the nitroen and Lewis structures.
See http: // ...
Bonds with single C atoms, 3 single CH bonds and one CN bond.
Entering N is a bit more complicated.
Each O atom has 6 valence electrons = 12.
N has 5 electrons plus 1 carbon atom = 6 electrons.
For a total of 18 electrons around 3 ONO atoms.
So put 4 pairs around each O atom.
These are just pairs of N 3 electrons, 1 for C and 1 for every O.
Therefore, N needs an extra pair.
As a general rule, this can be solved by converting 1 of the un-shared pairs of O to a common pair with N. Therefore, C, N and two O have 4 pairs of electrons.
Since the cycle in which O atoms move electrons is completely random, you can write two resonant structures for a molecule, and two with 1 and 1/2 bonds between molecule N and their corresponding ORs. The average of the structures.
Ch3no2 Lewis Structure
Ch3no2 Lewis Structure
Lewis structure of nitrone
Well, in the middle of the structure, C becomes N (CN).
There are three hydrogen atoms attached to the carbon atom (group IL). Two atoms of oxygen are attached to one nitrogen atom. The total number of valence electrons for this molecule is 24. When you factor in the bond pairs, there are 12 remaining electrons that need to be split.
Hydrogen requires only 2 valence electrons for each s, so it cannot add any remaining electrons to the hydrogen. In addition, the carbon atom is composed of four single bonds (three hydrogen atoms, one nitrogen atom). First, it divides electrons between nitrogen and oxygen into extremely negative electrons (oxygen). Each oxygen atom accepts 6 electrons.
The only problem is that nitrogen has only 6 electrons at this point (3 single bonds). To replenish nitrogen bytes, multiply one of the oxygen levels by nitrogen. For this molecule a resonant structure is formed, in which oxygen can bind nitrogen twice.
This page can help you.
D:
Lewis Diagram for Nitron, CH3NO2 !!!! Help?
Do your best to make the outline clear. Thank you very much