Ca C2h3o2 2 - How To Discuss

David Jones
Published May 08, 2026
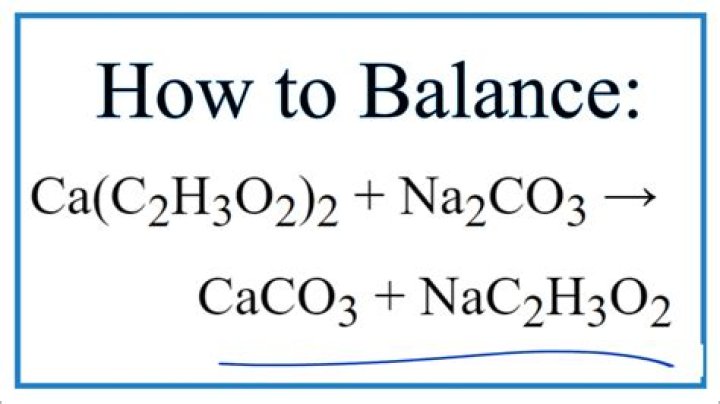
Ca C2h3o2 2
From the formula of calcium acetate, Ca (C2H3O2) 2? ۔
Calculate the carbon M obtained from 65.3 g of compound,
Calcium acetate molar m = 40 +4 (12) +6 (1) +16 (4) = 158.
Calcium acetate = 65.3 g / (158 g / mol) = 0.413
Experimental formula: (4 carbon / sesame of calcium acetate)
4 (0.413) = 1.653 mol c.
Grams C = 1.653 mol (12 g / mol) = 19.8 g carbon.
Acetate formula
Ca C2h3o2 2
Ca C2h3o2 2
From the formula of calcium acetate, Ca (C2H3O2) 2? 3
Calculate the carbon M obtained from 65.3 g of the mixture,
Calcium Acetate Mرller m = 40 + 4 (12) +6 (1) +16 (4) = 158
Calcium Acetate = 65.3 g / (158 g / mol) = 0.413
Experimental formula: (4 carbon / calcium acetate sesame)
4 (0.413) = 1.653 mol c
Grams C = 1.653 mol (12 g / mol) = 19.8 g carbon